Key Insights
The Canadian diabetes care devices market is poised for significant expansion, projected to reach approximately $1.93 million by 2025, exhibiting a robust Compound Annual Growth Rate (CAGR) of 4.98% through 2033. This growth is primarily fueled by an increasing prevalence of diabetes across Canada, driven by factors such as sedentary lifestyles, dietary shifts, and an aging population. Technological advancements in diabetes management devices are also playing a crucial role, with innovative insulin delivery systems and advanced glucose monitoring solutions becoming more accessible and sophisticated. The market is segmented into Management Devices, including insulin pumps, syringes, pens, cartridges, and jet injectors, and Monitoring Devices, encompassing self-monitoring blood glucose (SMBG) devices and continuous glucose monitoring (CGM) systems. The demand for these devices is directly linked to the growing need for effective and convenient diabetes management, empowering individuals to maintain better control over their blood glucose levels.
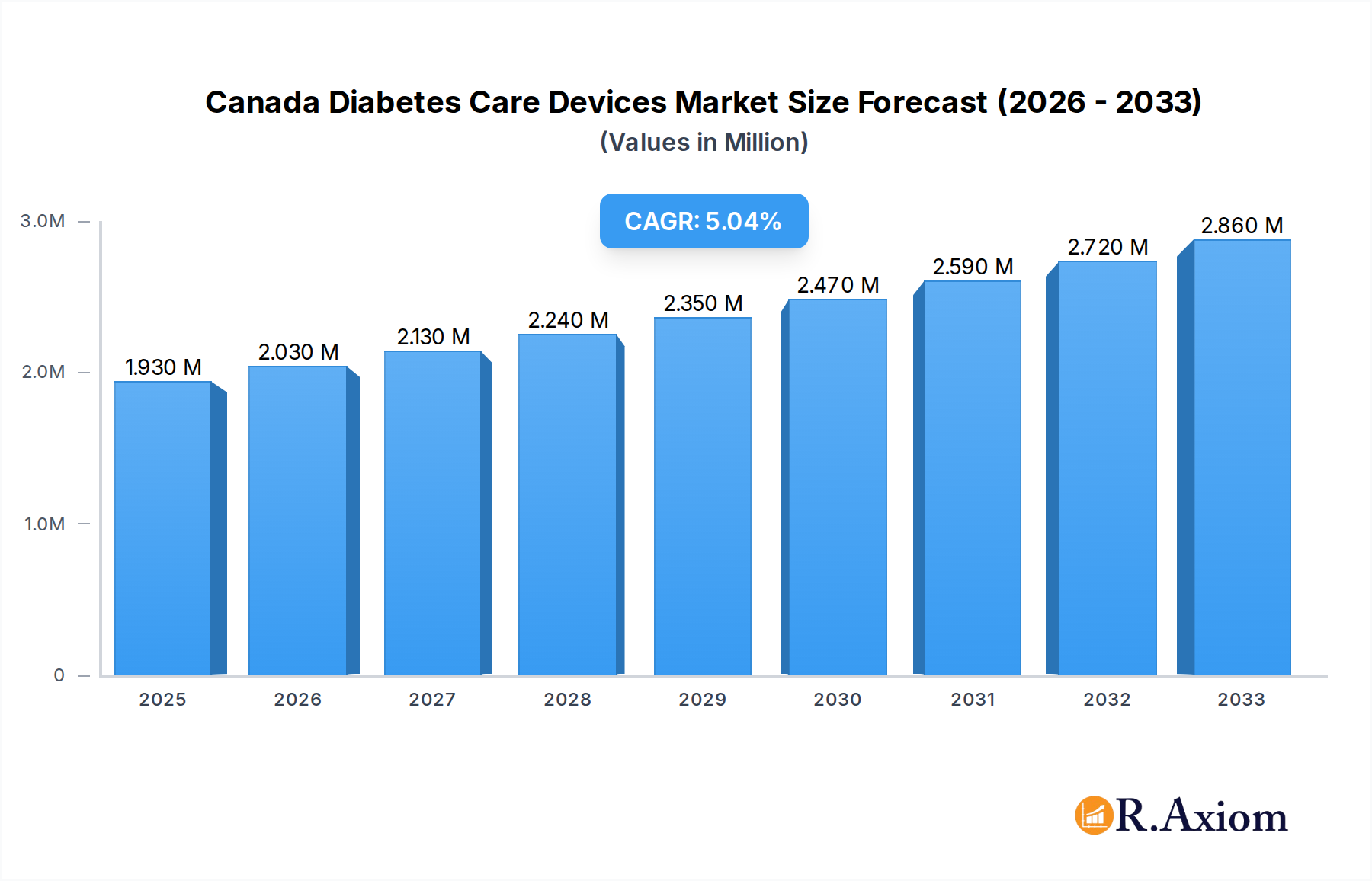
Canada Diabetes Care Devices Market Market Size (In Million)

Key trends shaping the Canadian diabetes care devices market include the rapid adoption of continuous glucose monitoring (CGM) technology, offering real-time data and reducing the need for frequent fingerstick tests. Insulin pumps are also gaining traction due to their ability to mimic the body's natural insulin delivery. While the market benefits from these positive drivers, it faces certain restraints, such as the high cost of advanced devices, which can limit accessibility for some patient segments, and reimbursement challenges. However, ongoing research and development by major players like Abbott Diabetes Care, Medtronic, Novo Nordisk, and Roche are focused on developing more affordable and user-friendly solutions. The increasing awareness among healthcare providers and patients about the benefits of early and consistent diabetes management is further accelerating market penetration, ensuring a positive trajectory for the Canadian diabetes care devices sector.
Canada Diabetes Care Devices Market Company Market Share

This in-depth report provides a detailed examination of the Canada Diabetes Care Devices Market, offering critical insights for industry stakeholders, investors, and manufacturers. Covering the historical period of 2019-2024 and projecting growth through 2033, this analysis delves into market dynamics, key players, emerging trends, and future opportunities. With a base year of 2025 and an estimated year also of 2025, the forecast period of 2025-2033 will capture significant market evolution. The report leverages high-traffic keywords such as "diabetes care devices Canada," "insulin pumps Canada," "CGM Canada," "blood glucose monitors Canada," and "diabetes management technology" to ensure maximum search visibility.
Canada Diabetes Care Devices Market Market Concentration & Innovation
The Canada Diabetes Care Devices Market exhibits a moderately concentrated landscape, with several global giants holding substantial market share alongside a growing number of innovative domestic players. Innovation is a key driver, fueled by advancements in sensor technology for continuous glucose monitoring (CGM) and the development of more user-friendly and automated insulin delivery systems. Regulatory frameworks, such as Health Canada approvals, play a crucial role in market entry and product adoption. While direct product substitutes are limited, lifestyle modifications and traditional diabetes management approaches represent indirect competition. End-user trends are increasingly leaning towards connected devices, remote monitoring capabilities, and personalized treatment plans. Mergers and acquisitions (M&A) are expected to continue as larger companies seek to integrate novel technologies and expand their product portfolios. For instance, the acquisition of smaller, specialized diabetes tech companies by established players can significantly alter market share. Anticipated M&A deal values in the Canadian market are projected to reach hundreds of millions of dollars as companies vie for technological leadership.
- Key Innovation Drivers:
- Miniaturization of devices
- Improved accuracy and reliability of sensors
- Enhanced data analytics and personalized insights
- Integration with telehealth platforms
- Development of needle-free injection technologies
- Regulatory Landscape:
- Health Canada approval processes for medical devices
- Reimbursement policies for diabetes care technologies
- Data privacy and security regulations (e.g., PIPEDA)
- Market Share Dynamics: Dominated by key players like Abbott Diabetes Care, Medtronic, and Dexcom, with significant contributions from Novo Nordisk A/S and Eli Lilly.
- M&A Trends: Focus on acquiring companies with cutting-edge CGM and insulin delivery technologies.
Canada Diabetes Care Devices Market Industry Trends & Insights
The Canada Diabetes Care Devices Market is experiencing robust growth, propelled by an increasing prevalence of diabetes across the nation, a growing awareness of the benefits of advanced diabetes management technologies, and supportive government initiatives. The compound annual growth rate (CAGR) for the market is estimated to be in the healthy range of 7-9% during the forecast period (2025-2033). Technological disruptions are at the forefront, with the rapid adoption of Continuous Glucose Monitoring (CGM) systems and the ongoing evolution of smart insulin pens and automated insulin delivery systems. Consumer preferences are shifting towards devices that offer greater convenience, accuracy, and the ability to proactively manage blood glucose levels, thereby reducing the risk of long-term complications. This includes a strong demand for connected devices that can sync with smartphones and provide real-time data to patients and healthcare providers. The competitive dynamics are intensifying, with companies differentiating themselves through product innovation, strategic partnerships, and expanded distribution networks. Market penetration for advanced diabetes care devices is steadily increasing, particularly among individuals with type 1 diabetes and those with poorly controlled type 2 diabetes. The rising incidence of obesity and sedentary lifestyles is also contributing to the growing diabetes burden, further driving the demand for effective diabetes management solutions.
- Market Growth Drivers:
- Rising diabetes prevalence (Type 1 and Type 2)
- Increasing healthcare expenditure on chronic disease management
- Growing adoption of advanced diabetes technologies (CGM, insulin pumps)
- Government initiatives and reimbursement policies for diabetes devices
- Aging population and associated rise in chronic diseases
- Technological Advancements:
- AI-powered diabetes management platforms
- Wearable biosensors for continuous monitoring
- Closed-loop insulin delivery systems
- Improved accuracy and usability of existing devices
- Consumer Preferences:
- Convenience and ease of use
- Accurate and real-time data
- Remote monitoring capabilities
- Integration with digital health ecosystems
- Personalized treatment insights
- Competitive Landscape:
- Intensifying competition among global and domestic players
- Focus on product differentiation and strategic alliances
- Increasing market share for CGM and insulin pump segments
Dominant Markets & Segments in Canada Diabetes Care Devices Market
The Monitoring Devices segment, particularly Continuous Glucose Monitoring (CGM) Devices, is emerging as a dominant force within the Canada Diabetes Care Devices Market. This segment is projected to witness the highest growth rate, driven by increasing awareness of its benefits in preventing hypoglycemia and hyperglycemia, improved patient outcomes, and expanding reimbursement coverage. The Self-monitoring Blood Glucose Devices segment, while mature, continues to hold a significant market share due to its accessibility and established user base.
Within the Management Devices segment, Insulin Pumps are experiencing substantial growth, reflecting a shift towards more sophisticated and automated insulin delivery solutions. The increasing adoption of these devices by individuals with type 1 diabetes and insulin-dependent type 2 diabetes patients is a key driver. Insulin Disposable Pens and Insulin Cartridges in Reusable pens remain crucial for a large portion of the insulin-using population, offering convenience and ease of use.
- Dominant Segments & Key Drivers:
- Continuous Glucose Monitoring (CGM) Devices:
- Driver: Enhanced glycemic control and reduction in complications.
- Driver: Expanding public and private insurance coverage.
- Driver: Technological advancements in sensor accuracy and wear time.
- Driver: Growing demand for real-time data for proactive management.
- Market Size: Estimated to reach over $500 Million by 2025.
- Self-monitoring Blood Glucose Devices:
- Driver: Cost-effectiveness and widespread availability.
- Driver: Continued reliance by a significant portion of the diabetic population.
- Driver: User-friendly interface and simple operation.
- Market Size: Estimated to be around $300 Million in 2025.
- Insulin Pumps:
- Driver: Improved quality of life and better glycemic variability management.
- Driver: Technological integration with CGM (hybrid closed-loop systems).
- Driver: Increasing physician recommendations for advanced insulin therapy.
- Market Size: Estimated to reach over $400 Million by 2025.
- Insulin Syringes:
- Driver: Affordability and established injection method.
- Driver: Preferred by some users for flexibility in insulin dosing.
- Market Size: Estimated to be around $150 Million in 2025.
- Insulin Disposable Pens & Insulin Cartridges in Reusable pens:
- Driver: Convenience and reduced injection complexity.
- Driver: Portability and discreetness.
- Driver: Variety of pen types catering to different user needs.
- Market Size: Combined estimated at over $350 Million in 2025.
- Insulin Jet Injectors:
- Driver: Needle-free administration appeals to needle-phobic patients.
- Driver: Potential for improved insulin absorption.
- Market Size: Smaller segment, estimated at $50 Million in 2025, with growth potential.
- Continuous Glucose Monitoring (CGM) Devices:
Canada Diabetes Care Devices Market Product Developments
Product developments in the Canada Diabetes Care Devices Market are heavily focused on enhancing user experience, improving data accuracy, and fostering greater integration between devices. Innovations in Continuous Glucose Monitoring (CGM) include longer wear times for sensors, improved accuracy even during rapid glucose fluctuations, and smaller, more discreet sensor designs. For insulin delivery, advancements are seen in the development of smart insulin pens that track doses and integrate with mobile apps, and sophisticated insulin pumps that offer hybrid closed-loop systems, automating insulin delivery based on CGM readings. The trend towards needle-free injection systems, such as NuGen Medical Devices' InsuJet approved by Health Canada in July 2022, signals a significant shift towards patient comfort and adherence. These product developments aim to empower individuals with diabetes to achieve better glycemic control with less effort and fewer complications, providing a competitive advantage to companies investing in cutting-edge research and development.
Report Scope & Segmentation Analysis
This report meticulously analyzes the Canada Diabetes Care Devices Market, segmented into two primary categories: Management Devices and Monitoring Devices. The Management Devices segment encompasses Insulin Pump, Insulin Syringes, Insulin Disposable Pens, Insulin Cartridges in Reusable pens, and Insulin Jet Injectors. The Monitoring Devices segment includes Self-monitoring Blood Glucose Devices and Continuous Glucose Monitoring. Each segment is analyzed for its current market size, projected growth, key market drivers, competitive landscape, and technological advancements during the forecast period of 2025-2033.
- Management Devices: This comprehensive category is projected to grow steadily, with insulin pumps leading the charge due to advanced automation. Insulin syringes, while established, will see a slower growth trajectory. Disposable pens and cartridges offer convenience and will maintain a significant market share. Insulin jet injectors represent an emerging niche with significant potential for adoption.
- Monitoring Devices: This segment is expected to be the fastest-growing, driven by the widespread adoption of CGM. Self-monitoring blood glucose devices will continue to be a cornerstone due to their affordability and established user base. The integration of both types of monitoring devices with management systems is a key trend.
Key Drivers of Canada Diabetes Care Devices Market Growth
The Canada Diabetes Care Devices Market is propelled by a confluence of powerful growth drivers. The escalating prevalence of diabetes, both type 1 and type 2, directly correlates with an increased demand for effective management tools. Government initiatives, such as expanded public coverage for advanced technologies like CGM through programs like Ontario's Assistive Devices Program (ADP) as seen in March 2022 for the Dexcom G6, significantly boost market penetration. Technological innovation, exemplified by advancements in CGM accuracy and the development of automated insulin delivery systems, plays a pivotal role in driving adoption. Furthermore, growing patient awareness regarding the long-term health benefits of proactive diabetes management, including reduced complication rates, fuels the demand for sophisticated devices.
- Rising Diabetes Prevalence: Canada faces a growing burden of diabetes, necessitating a greater need for management solutions.
- Government Reimbursement Policies: Increased coverage for devices like CGM improves affordability and accessibility.
- Technological Advancements: Innovations in accuracy, connectivity, and automation drive product adoption.
- Patient Awareness: Growing understanding of the benefits of proactive and data-driven diabetes management.
Challenges in the Canada Diabetes Care Devices Market Sector
Despite the promising growth trajectory, the Canada Diabetes Care Devices Market faces several challenges. High upfront costs for some advanced devices, such as insulin pumps and continuous glucose monitors, can be a barrier to widespread adoption, especially for individuals without comprehensive insurance coverage. Navigating the complex regulatory approval processes for new devices can be time-consuming and resource-intensive for manufacturers. Furthermore, ensuring consistent supply chain management and addressing potential shortages of critical components can impact market availability. The need for continuous patient education and training on the effective use of these technologies also presents an ongoing challenge. Intense competition among established players and emerging innovators can also lead to pricing pressures.
- High Device Costs: Affordability remains a concern for certain advanced technologies.
- Regulatory Hurdles: Stringent approval processes can delay market entry.
- Supply Chain Disruptions: Ensuring consistent availability of devices and components.
- Patient Education and Training: Crucial for effective utilization and adherence.
- Reimbursement Variations: Inconsistent coverage across different provinces and insurance plans.
Emerging Opportunities in Canada Diabetes Care Devices Market
The Canada Diabetes Care Devices Market is ripe with emerging opportunities, driven by technological advancements and evolving patient needs. The integration of Artificial Intelligence (AI) and Machine Learning (ML) into diabetes management platforms offers predictive analytics for glycemic control and personalized treatment recommendations, creating a significant opportunity for smart device development. The expansion of telehealth and remote patient monitoring services presents a lucrative avenue for connected diabetes devices that can transmit data seamlessly to healthcare providers. As public and private payers increasingly recognize the cost-effectiveness of advanced diabetes care in preventing long-term complications, there is a growing opportunity for expanded reimbursement coverage. Furthermore, the development of user-friendly, integrated systems that simplify the diabetes management experience for patients of all ages, including pediatric populations, will be crucial for capturing market share. The growing interest in preventative health and wellness also opens doors for early detection and intervention devices.
- AI and Machine Learning Integration: Predictive analytics and personalized treatment plans.
- Telehealth and Remote Monitoring: Connected devices for seamless data transmission.
- Expanded Reimbursement Coverage: Growing recognition of cost-effectiveness of advanced care.
- Pediatric and Geriatric Solutions: Tailored devices for specific age groups.
- Preventative Health Technologies: Early detection and intervention solutions.
Leading Players in the Canada Diabetes Care Devices Market Market
- AgaMatrix Inc.
- Novo Nordisk A/S
- Eli Lilly
- Abbott Diabetes Care
- Medtronic
- Ypsomed Holding AG
- Sanofi Aventis
- F Hoffmann-La Roche Ltd
- Lifescan
- Terumo
- Arkray
- Dexcom
- Ascensia Diabetes Care
Key Developments in Canada Diabetes Care Devices Market Industry
- July 2022: NuGen Medical Devices Inc. announced that its needle-free injection system and accessories, also known as InsuJet, were approved by Health Canada specifically for needle-free insulin delivery.
- March 2022: The Ontario government announced that it would provide coverage for the Dexcom G6 CGM System through Ontario's Assistive Devices Program (ADP) for people in the province with type 1 diabetes who are over 2 years of age and meet coverage criteria. The Non-Insured health benefits program also covers first nations and Inuit children. With expanded public coverage for CGM, more people can access this standard care technology, helping them to manage life-long chronic illnesses more effectively.
Strategic Outlook for Canada Diabetes Care Devices Market Market
The strategic outlook for the Canada Diabetes Care Devices Market is exceptionally positive, driven by continuous innovation and increasing adoption of advanced technologies. Companies that focus on developing integrated ecosystems, offering seamless data flow between monitoring and management devices, will likely gain a competitive edge. Strategic partnerships with healthcare providers, insurance companies, and patient advocacy groups will be crucial for market expansion and navigating reimbursement landscapes. Furthermore, investing in user-centric design and robust customer support will foster patient loyalty and adherence. The growing demand for personalized medicine in diabetes care presents an opportunity for companies to leverage data analytics to offer tailored solutions. Ultimately, the market's trajectory will be shaped by a commitment to improving patient outcomes, enhancing convenience, and driving technological advancements in the fight against diabetes.
Canada Diabetes Care Devices Market Segmentation
-
1. Management Devices
- 1.1. Insulin Pump
- 1.2. Insulin Syringes
- 1.3. Insulin Disposable Pens
- 1.4. Insulin Cartridges in Reusable pens
- 1.5. Insulin Jet Injectors
-
2. Monitoring Devices
- 2.1. Self-monitoring Blood Glucose Devices
- 2.2. Continuous Glucose Monitoring
Canada Diabetes Care Devices Market Segmentation By Geography
- 1. Canada
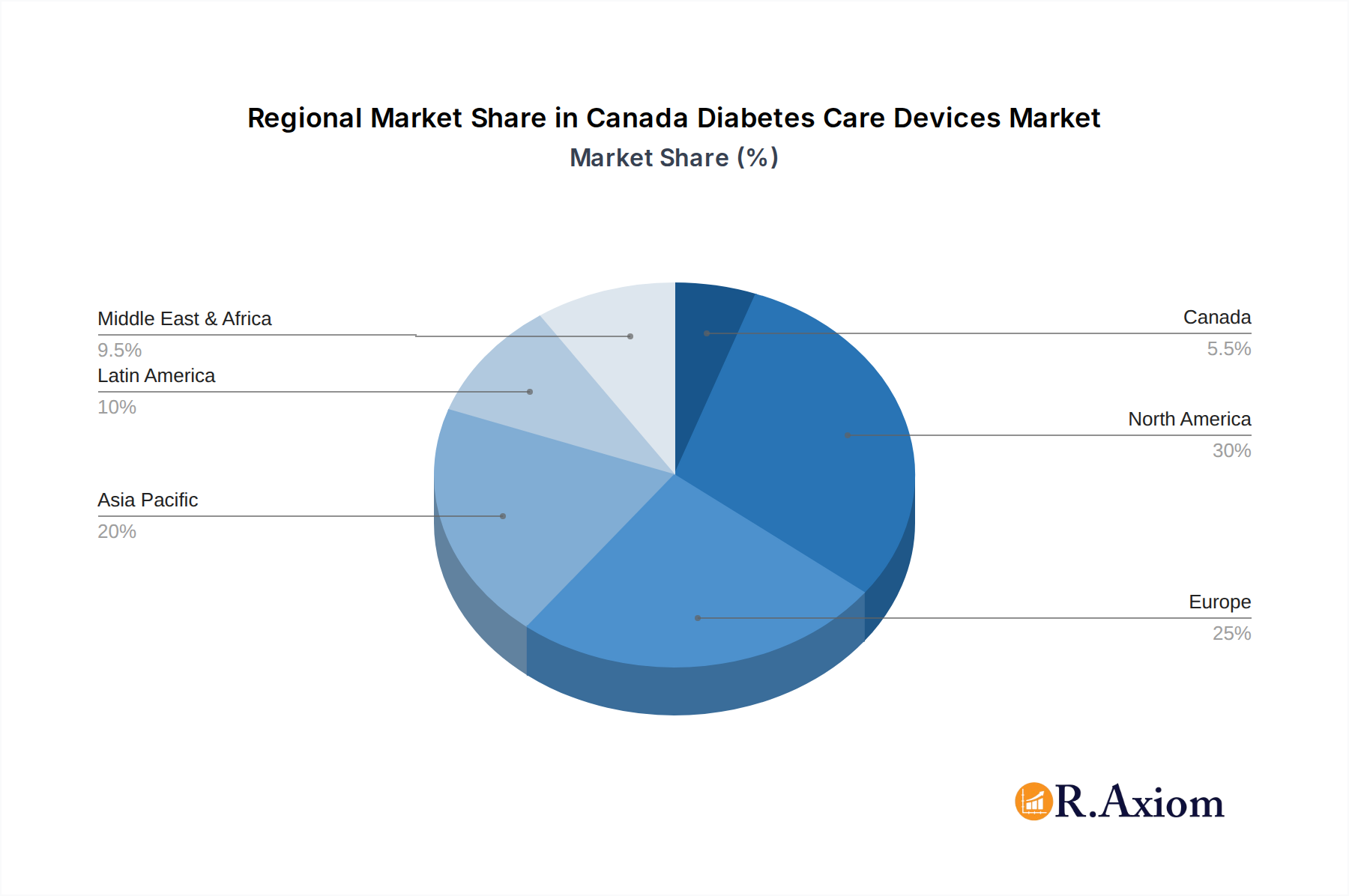
Canada Diabetes Care Devices Market Regional Market Share

Geographic Coverage of Canada Diabetes Care Devices Market
Canada Diabetes Care Devices Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 4.98% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. RAX Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Management Devices
- 5.1.1. Insulin Pump
- 5.1.2. Insulin Syringes
- 5.1.3. Insulin Disposable Pens
- 5.1.4. Insulin Cartridges in Reusable pens
- 5.1.5. Insulin Jet Injectors
- 5.2. Market Analysis, Insights and Forecast - by Monitoring Devices
- 5.2.1. Self-monitoring Blood Glucose Devices
- 5.2.2. Continuous Glucose Monitoring
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. Canada
- 5.1. Market Analysis, Insights and Forecast - by Management Devices
- 6. Canada Diabetes Care Devices Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Management Devices
- 6.1.1. Insulin Pump
- 6.1.2. Insulin Syringes
- 6.1.3. Insulin Disposable Pens
- 6.1.4. Insulin Cartridges in Reusable pens
- 6.1.5. Insulin Jet Injectors
- 6.2. Market Analysis, Insights and Forecast - by Monitoring Devices
- 6.2.1. Self-monitoring Blood Glucose Devices
- 6.2.2. Continuous Glucose Monitoring
- 6.1. Market Analysis, Insights and Forecast - by Management Devices
- 7. Competitive Analysis
- 7.1. Company Profiles
- 7.1.1 AgaMatrix Inc
- 7.1.1.1. Company Overview
- 7.1.1.2. Products
- 7.1.1.3. Company Financials
- 7.1.1.4. SWOT Analysis
- 7.1.2 2 Novo Nordisk A/S
- 7.1.2.1. Company Overview
- 7.1.2.2. Products
- 7.1.2.3. Company Financials
- 7.1.2.4. SWOT Analysis
- 7.1.3 Insulin Devices
- 7.1.3.1. Company Overview
- 7.1.3.2. Products
- 7.1.3.3. Company Financials
- 7.1.3.4. SWOT Analysis
- 7.1.4 Eli Lilly
- 7.1.4.1. Company Overview
- 7.1.4.2. Products
- 7.1.4.3. Company Financials
- 7.1.4.4. SWOT Analysis
- 7.1.5 Abbott Diabetes Care
- 7.1.5.1. Company Overview
- 7.1.5.2. Products
- 7.1.5.3. Company Financials
- 7.1.5.4. SWOT Analysis
- 7.1.6 Medtronic
- 7.1.6.1. Company Overview
- 7.1.6.2. Products
- 7.1.6.3. Company Financials
- 7.1.6.4. SWOT Analysis
- 7.1.7 Ypsomed Holding AG
- 7.1.7.1. Company Overview
- 7.1.7.2. Products
- 7.1.7.3. Company Financials
- 7.1.7.4. SWOT Analysis
- 7.1.8 Continuous Glucose Monitoring Devices
- 7.1.8.1. Company Overview
- 7.1.8.2. Products
- 7.1.8.3. Company Financials
- 7.1.8.4. SWOT Analysis
- 7.1.9 1 Abbott Diabetes Care
- 7.1.9.1. Company Overview
- 7.1.9.2. Products
- 7.1.9.3. Company Financials
- 7.1.9.4. SWOT Analysis
- 7.1.10 Sanofi Aventis
- 7.1.10.1. Company Overview
- 7.1.10.2. Products
- 7.1.10.3. Company Financials
- 7.1.10.4. SWOT Analysis
- 7.1.11 2 Medtronic PLC
- 7.1.11.1. Company Overview
- 7.1.11.2. Products
- 7.1.11.3. Company Financials
- 7.1.11.4. SWOT Analysis
- 7.1.12 2 F Hoffmann-La Roche Ltd
- 7.1.12.1. Company Overview
- 7.1.12.2. Products
- 7.1.12.3. Company Financials
- 7.1.12.4. SWOT Analysis
- 7.1.13 Lifescan
- 7.1.13.1. Company Overview
- 7.1.13.2. Products
- 7.1.13.3. Company Financials
- 7.1.13.4. SWOT Analysis
- 7.1.14 Self-monitoring Blood Glucose Devices
- 7.1.14.1. Company Overview
- 7.1.14.2. Products
- 7.1.14.3. Company Financials
- 7.1.14.4. SWOT Analysis
- 7.1.15 Novo Nordisk A/S
- 7.1.15.1. Company Overview
- 7.1.15.2. Products
- 7.1.15.3. Company Financials
- 7.1.15.4. SWOT Analysis
- 7.1.16 Terumo
- 7.1.16.1. Company Overview
- 7.1.16.2. Products
- 7.1.16.3. Company Financials
- 7.1.16.4. SWOT Analysis
- 7.1.17 Arkray
- 7.1.17.1. Company Overview
- 7.1.17.2. Products
- 7.1.17.3. Company Financials
- 7.1.17.4. SWOT Analysis
- 7.1.18 1 Medtronic PLC
- 7.1.18.1. Company Overview
- 7.1.18.2. Products
- 7.1.18.3. Company Financials
- 7.1.18.4. SWOT Analysis
- 7.1.19 3 Dexcom
- 7.1.19.1. Company Overview
- 7.1.19.2. Products
- 7.1.19.3. Company Financials
- 7.1.19.4. SWOT Analysis
- 7.1.20 Dexcom
- 7.1.20.1. Company Overview
- 7.1.20.2. Products
- 7.1.20.3. Company Financials
- 7.1.20.4. SWOT Analysis
- 7.1.21 Ascensia Diabetes Care
- 7.1.21.1. Company Overview
- 7.1.21.2. Products
- 7.1.21.3. Company Financials
- 7.1.21.4. SWOT Analysis
- 7.1.22 F Hoffmann-La Roche
- 7.1.22.1. Company Overview
- 7.1.22.2. Products
- 7.1.22.3. Company Financials
- 7.1.22.4. SWOT Analysis
- 7.1.1 AgaMatrix Inc
- 7.2. Market Entropy
- 7.2.1 Company's Key Areas Served
- 7.2.2 Recent Developments
- 7.3. Company Market Share Analysis 2025
- 7.3.1 Top 5 Companies Market Share Analysis
- 7.3.2 Top 3 Companies Market Share Analysis
- 7.4. List of Potential Customers
- 8. Research Methodology
List of Figures
- Figure 1: Canada Diabetes Care Devices Market Revenue Breakdown (Million, %) by Product 2025 & 2033
- Figure 2: Canada Diabetes Care Devices Market Share (%) by Company 2025
List of Tables
- Table 1: Canada Diabetes Care Devices Market Revenue Million Forecast, by Management Devices 2020 & 2033
- Table 2: Canada Diabetes Care Devices Market Volume K Unit Forecast, by Management Devices 2020 & 2033
- Table 3: Canada Diabetes Care Devices Market Revenue Million Forecast, by Monitoring Devices 2020 & 2033
- Table 4: Canada Diabetes Care Devices Market Volume K Unit Forecast, by Monitoring Devices 2020 & 2033
- Table 5: Canada Diabetes Care Devices Market Revenue Million Forecast, by Region 2020 & 2033
- Table 6: Canada Diabetes Care Devices Market Volume K Unit Forecast, by Region 2020 & 2033
- Table 7: Canada Diabetes Care Devices Market Revenue Million Forecast, by Management Devices 2020 & 2033
- Table 8: Canada Diabetes Care Devices Market Volume K Unit Forecast, by Management Devices 2020 & 2033
- Table 9: Canada Diabetes Care Devices Market Revenue Million Forecast, by Monitoring Devices 2020 & 2033
- Table 10: Canada Diabetes Care Devices Market Volume K Unit Forecast, by Monitoring Devices 2020 & 2033
- Table 11: Canada Diabetes Care Devices Market Revenue Million Forecast, by Country 2020 & 2033
- Table 12: Canada Diabetes Care Devices Market Volume K Unit Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Canada Diabetes Care Devices Market?
The projected CAGR is approximately 4.98%.
2. Which companies are prominent players in the Canada Diabetes Care Devices Market?
Key companies in the market include AgaMatrix Inc, 2 Novo Nordisk A/S, Insulin Devices, Eli Lilly, Abbott Diabetes Care, Medtronic, Ypsomed Holding AG, Continuous Glucose Monitoring Devices, 1 Abbott Diabetes Care, Sanofi Aventis, 2 Medtronic PLC, 2 F Hoffmann-La Roche Ltd, Lifescan, Self-monitoring Blood Glucose Devices, Novo Nordisk A/S, Terumo, Arkray, 1 Medtronic PLC, 3 Dexcom, Dexcom, Ascensia Diabetes Care, F Hoffmann-La Roche.
3. What are the main segments of the Canada Diabetes Care Devices Market?
The market segments include Management Devices, Monitoring Devices.
4. Can you provide details about the market size?
The market size is estimated to be USD 1.93 Million as of 2022.
5. What are some drivers contributing to market growth?
; The Rise in Global Prevalence of Cases of Obesity due to Modern Sedentary Lifestyles; Rise in Awareness and Disposable Income in Developed Economies.
6. What are the notable trends driving market growth?
Rising Diabetes Prevalence in Canada.
7. Are there any restraints impacting market growth?
; Highly Cost of Branded Products in Emerging Countries; Severe Adverse Associated with Medication Including Seizures. Suicidal Attempts and Even Death; Adoption of Traditional Yoga and Herbal Products.
8. Can you provide examples of recent developments in the market?
July 2022: NuGen Medical Devices Inc. announced that its needle-free injection system and accessories, also known as InsuJet, were approved by Health Canada specifically for needle-free insulin delivery.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3800, USD 4500, and USD 5800 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Canada Diabetes Care Devices Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Canada Diabetes Care Devices Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Canada Diabetes Care Devices Market?
To stay informed about further developments, trends, and reports in the Canada Diabetes Care Devices Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
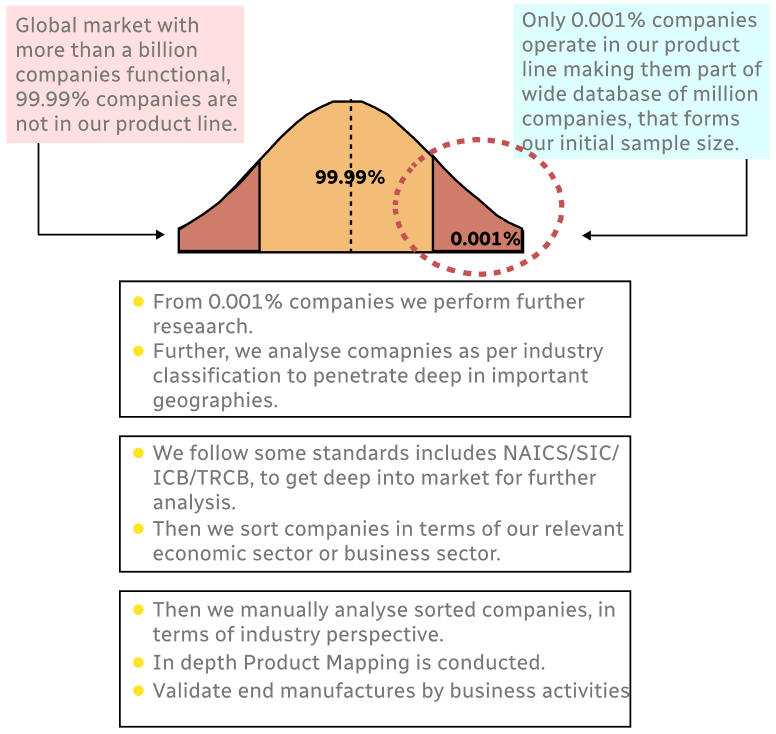
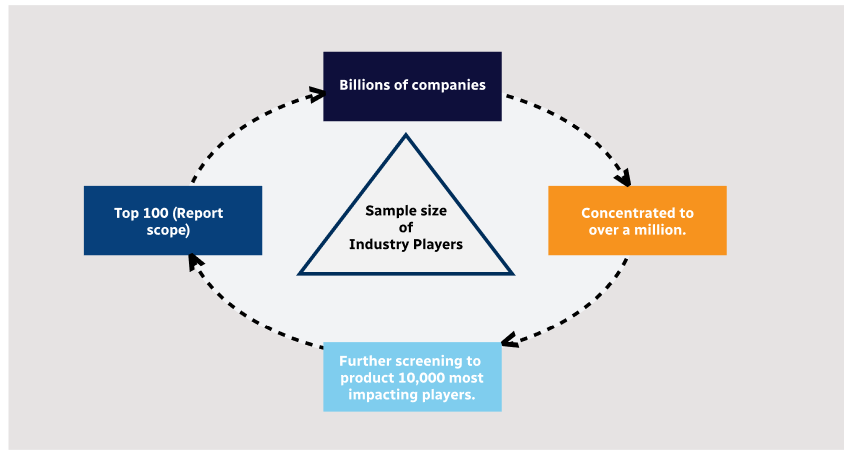
Step 1 - Identification of Relevant Samples Size from Population Database
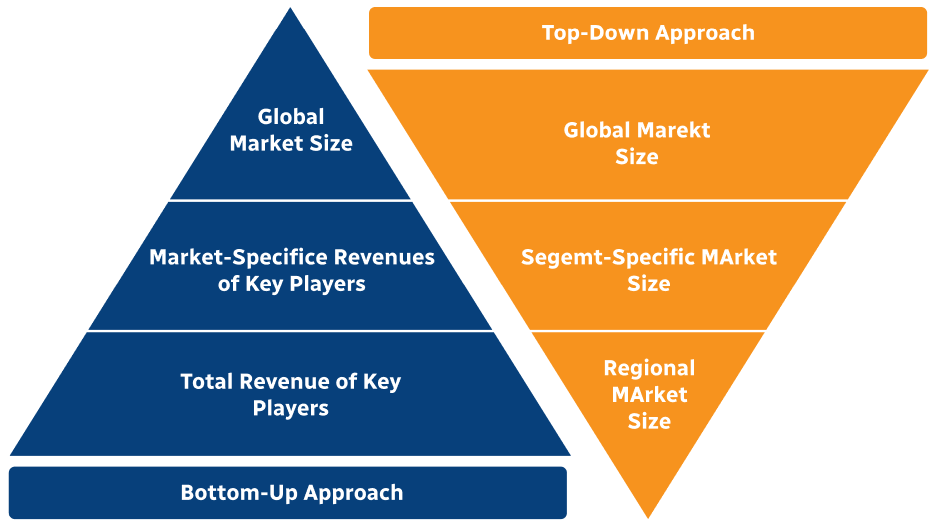
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence