Key Insights
The global Rare Disease Treatment Market is poised for substantial expansion, projected to reach an estimated $216.65 million by 2025. This growth is fueled by a compelling compound annual growth rate (CAGR) of 11.93%, indicating a dynamic and rapidly evolving landscape. A primary driver for this surge is the increasing understanding and diagnosis of rare diseases, coupled with significant advancements in scientific research and therapeutic development. The market is characterized by a strong focus on biologics, which represent a significant portion of the drug type segment due to their efficacy in treating complex genetic and autoimmune rare conditions. Therapeutic areas like genetic diseases and oncology are leading the charge, attracting substantial investment and innovation. The growing pipeline of targeted therapies, particularly for conditions with high unmet medical needs, is a key factor propelling market growth. Furthermore, greater patient advocacy and improved healthcare infrastructure in developed and emerging economies are enhancing access to specialized treatments, contributing to the overall market valuation.
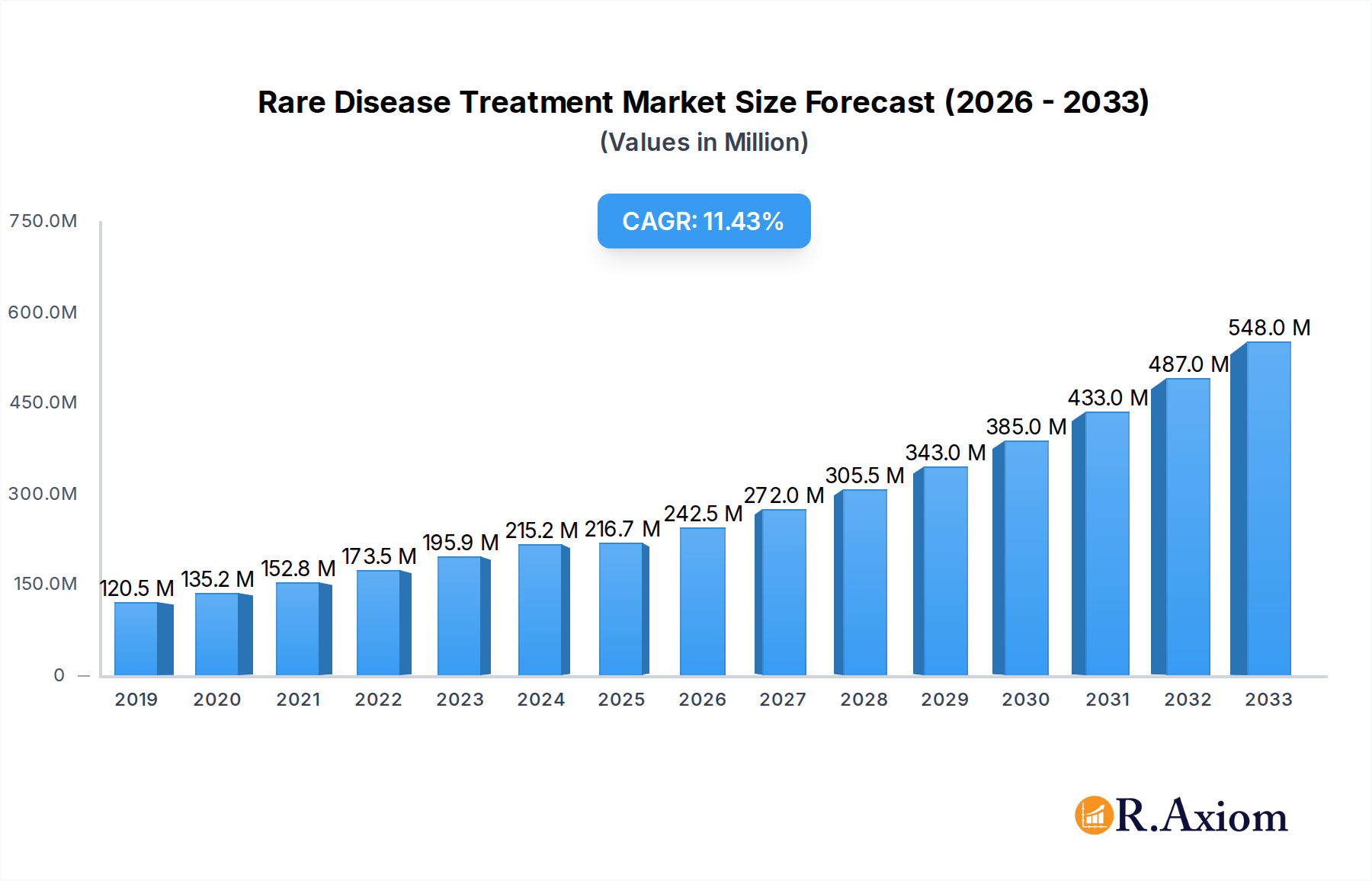
Rare Disease Treatment Market Market Size (In Million)

The market's trajectory is further shaped by key trends such as the rise of precision medicine, which allows for more tailored and effective treatment strategies for rare genetic disorders. Technological innovations in drug delivery systems, including advanced oral formulations and sophisticated injection methods, are also enhancing patient compliance and treatment outcomes. However, challenges such as high research and development costs, the complexities of clinical trials for small patient populations, and pricing pressures remain important considerations. Despite these restraints, the expanding pipeline of novel therapies, strategic collaborations between pharmaceutical giants and smaller biotech firms, and supportive regulatory frameworks for orphan drugs are expected to sustain the robust growth momentum. North America and Europe currently dominate the market due to established healthcare systems and high awareness levels, but the Asia Pacific region is emerging as a significant growth frontier, driven by increasing healthcare expenditure and a growing incidence of rare diseases.
Rare Disease Treatment Market Company Market Share

This in-depth report provides a meticulous examination of the global Rare Disease Treatment Market, offering critical insights and actionable intelligence for stakeholders navigating this complex and rapidly evolving landscape. The study covers the historical period from 2019 to 2024, the base year of 2025, and extends through an extensive forecast period from 2025 to 2033. With a projected market size of XX Million by 2025 and a robust Compound Annual Growth Rate (CAGR) of XX%, this report is an indispensable resource for pharmaceutical companies, biotech innovators, investors, regulatory bodies, and patient advocacy groups.
Rare Disease Treatment Market Market Concentration & Innovation
The Rare Disease Treatment Market exhibits a moderate to high degree of concentration, driven by the specialized nature of research, development, and manufacturing. Key players like Pfizer Inc, AstraZeneca (Alexion Pharmaceuticals Inc), Novartis AG, and Amgen Inc dominate significant market share segments due to their established pipelines and extensive expertise in rare disease therapeutics. Innovation is a paramount driver, fueled by advancements in genetic sequencing, gene editing technologies (CRISPR-Cas9), and personalized medicine approaches. These innovations are crucial for addressing unmet medical needs in a segment where traditional drug development models often fall short. Regulatory frameworks, particularly those from the FDA and EMA, play a pivotal role, often offering incentives like orphan drug designations and expedited review pathways to encourage development. The presence of product substitutes is relatively limited due to the highly specific nature of rare diseases, making novel therapeutic interventions highly sought after. End-user trends lean towards treatments offering improved efficacy, reduced side effects, and better quality of life, with a growing emphasis on patient-centric care models. Mergers and acquisitions (M&A) remain a strategic tool for market consolidation and pipeline expansion, with significant deal values often seen as companies seek to acquire promising early-stage or late-stage assets. For instance, acquisitions of smaller biotech firms with novel rare disease platforms are common. The overall market share distribution indicates that the top 5-10 companies collectively hold over 60% of the market revenue. M&A deal values in the rare disease space have consistently exceeded XX Million in recent years, reflecting the high potential return on investment.
Rare Disease Treatment Market Industry Trends & Insights
The Rare Disease Treatment Market is experiencing unprecedented growth, propelled by a confluence of scientific breakthroughs, enhanced diagnostic capabilities, and increasing global awareness of rare conditions. The market is projected to reach XX Million by 2025, with a projected CAGR of XX% over the forecast period (2025–2033). This robust growth is underpinned by significant investments in research and development, spurred by the potential for high therapeutic impact and favorable regulatory incentives. Technological disruptions, particularly in the realm of gene therapy, cell therapy, and precision medicine, are revolutionizing treatment paradigms. These advanced modalities offer the prospect of addressing the root causes of rare genetic disorders, moving beyond symptomatic management. For example, the increasing success of gene replacement therapies for conditions like Spinal Muscular Atrophy (SMA) underscores the transformative potential. Consumer preferences are increasingly shifting towards treatments that offer long-term solutions and significantly improve patients' quality of life, driving demand for innovative and effective therapies. The competitive dynamics are characterized by a blend of established pharmaceutical giants and agile biotech startups, each vying for a position in this lucrative and impactful market. Collaboration between academic institutions, pharmaceutical companies, and patient advocacy groups is becoming increasingly common, fostering a more efficient R&D ecosystem. The market penetration of advanced therapies, while still nascent for many rare diseases, is expected to surge as regulatory hurdles are navigated and manufacturing scales improve. The increasing availability of genomic data and advanced diagnostic tools is also contributing to earlier and more accurate diagnoses, thereby expanding the eligible patient population for existing and pipeline treatments. The industry is witnessing a paradigm shift from a one-size-fits-all approach to highly personalized treatment strategies, tailored to the specific genetic profile of individual patients. This trend is further amplified by the growing understanding of disease heterogeneity within rare conditions, demanding bespoke therapeutic interventions.
Dominant Markets & Segments in Rare Disease Treatment Market
The Rare Disease Treatment Market is shaped by diverse regional dynamics, therapeutic areas, drug types, and administration modes.
Leading Regions: North America, particularly the United States, currently dominates the market, driven by a robust healthcare infrastructure, substantial R&D investments, and favorable regulatory support for orphan drugs. Europe, with its strong pharmaceutical presence and a growing number of rare disease initiatives, is the second-largest market. Asia-Pacific is emerging as a significant growth region due to increasing healthcare expenditure, improving diagnostic capabilities, and a rising prevalence of certain rare diseases.
- Key Drivers in North America: High disposable income, advanced medical technology, and strong patient advocacy groups are key economic and social drivers. Favorable reimbursement policies and government initiatives like the Orphan Drug Act have significantly bolstered market growth.
- Key Drivers in Europe: Harmonized regulatory pathways (EMA), national rare disease plans, and increasing collaboration among member states contribute to market expansion.
Dominant Therapeutic Areas:
- Genetic Diseases: This segment holds a substantial share due to the inherent genetic basis of many rare conditions. Therapies targeting rare blood disorders, lysosomal storage diseases, and inherited metabolic disorders are particularly prominent.
- Key Drivers: Breakthroughs in gene therapy and enzyme replacement therapies, coupled with a high unmet need.
- Neurological Diseases: Conditions like Huntington's disease, Amyotrophic Lateral Sclerosis (ALS), and rare forms of epilepsy represent a significant, albeit challenging, therapeutic area with ongoing research and development efforts.
- Key Drivers: Growing understanding of neurological pathways and the development of targeted therapies.
- Oncology: While not exclusively a rare disease segment, certain rare cancers and subtypes of common cancers with rare genetic mutations fall under this umbrella.
- Key Drivers: Advances in targeted therapies and immunotherapy are expanding treatment options.
- Infectious Diseases: Rare infectious diseases, often with limited treatment options, also contribute to the market.
- Key Drivers: Emergence of novel pathogens and the need for specialized treatments.
- Cardiovascular Diseases: Rare forms of congenital heart defects and cardiomyopathies are addressed within this segment.
- Key Drivers: Improved diagnostic tools and surgical interventions.
- Genetic Diseases: This segment holds a substantial share due to the inherent genetic basis of many rare conditions. Therapies targeting rare blood disorders, lysosomal storage diseases, and inherited metabolic disorders are particularly prominent.
Dominant Drug Types:
- Biologics: This segment is a major growth engine, encompassing monoclonal antibodies, recombinant proteins, and gene/cell therapies. Their high specificity and efficacy in treating complex rare diseases drive their dominance.
- Key Drivers: Technological advancements in protein engineering and genetic manipulation, offering highly targeted treatments.
- Non-biologics: Small molecule drugs continue to play a role, particularly in managing symptoms and providing alternative treatment options for certain rare conditions.
- Key Drivers: Established manufacturing processes and cost-effectiveness for some indications.
- Biologics: This segment is a major growth engine, encompassing monoclonal antibodies, recombinant proteins, and gene/cell therapies. Their high specificity and efficacy in treating complex rare diseases drive their dominance.
Dominant Modes of Administration:
- Injection: Intravenous and subcutaneous injections are prevalent, especially for biologics and enzyme replacement therapies requiring precise dosing and systemic delivery.
- Key Drivers: Essential for delivering complex biologics and ensuring systemic availability.
- Oral: Oral formulations are gaining traction for non-biologics and some newer oral biologics, offering greater patient convenience and adherence.
- Key Drivers: Patient preference for ease of administration and improved adherence.
- Other Modes of Administration: This includes topical applications, inhalation, and intrathecal delivery, which are crucial for specific rare diseases affecting localized areas or the central nervous system.
- Key Drivers: Tailored delivery mechanisms for specific disease targets.
- Injection: Intravenous and subcutaneous injections are prevalent, especially for biologics and enzyme replacement therapies requiring precise dosing and systemic delivery.
Rare Disease Treatment Market Product Developments
Product development in the rare disease treatment market is characterized by a relentless pursuit of innovative therapies that address unmet needs. Companies are focusing on advanced modalities like gene therapies and cell therapies, offering the potential for one-time curative treatments for genetic disorders. Enzyme replacement therapies (ERTs) and protein therapies continue to evolve, with improved efficacy and delivery mechanisms. The development of personalized medicines, tailored to an individual's genetic makeup, is a significant trend, enhancing treatment efficacy and minimizing side effects. These product developments are driven by cutting-edge research in molecular biology, genomics, and drug delivery systems, aiming to provide life-changing interventions for patients with previously untreatable conditions. The competitive advantage lies in early market entry, robust clinical trial data, and strong intellectual property protection.
Report Scope & Segmentation Analysis
This comprehensive report meticulously segments the Rare Disease Treatment Market across key parameters, providing granular insights into each area. The segmentation includes:
- Drug Type:
- Biologics: This segment encompasses a wide range of advanced therapies including monoclonal antibodies, recombinant proteins, gene therapies, and cell therapies. These are crucial for treating complex genetic and autoimmune rare diseases.
- Non-biologics: This includes traditional small molecule drugs and generics that offer symptomatic relief or disease management for specific rare conditions.
- Therapeutic Area:
- Genetic Diseases: This forms a substantial segment, covering inherited metabolic disorders, lysosomal storage diseases, blood disorders, and rare syndromes.
- Neurological Diseases: This segment addresses rare neurological conditions such as muscular dystrophies, rare epilepsies, and neurodegenerative disorders.
- Oncology: This includes rare cancers and specific rare subtypes of more common cancers.
- Infectious Diseases: This segment covers rare and neglected tropical diseases.
- Cardiovascular Diseases: This addresses rare congenital heart defects and inherited cardiac conditions.
- Other Therapeutic Area: This encompasses a broad range of rare conditions not falling into the above categories, such as rare dermatological and ophthalmological disorders.
- Mode of Administration:
- Oral: This segment includes pills, capsules, and liquid formulations, favored for their convenience.
- Injection: This covers intravenous, subcutaneous, and intramuscular routes, essential for many biologics.
- Other Modes of Administration: This includes topical, inhalation, intrathecal, and other specialized delivery methods tailored to specific diseases.
Each segment is analyzed for its current market size, projected growth trajectory, and competitive landscape.
Key Drivers of Rare Disease Treatment Market Growth
The Rare Disease Treatment Market is propelled by a potent mix of factors. Significant advancements in genetic sequencing and diagnostic technologies enable earlier and more accurate identification of rare diseases, thereby expanding the patient pool and demand for treatments. Favorable regulatory landscapes, including orphan drug designations and expedited review pathways in major markets like the US and EU, incentivize research and development by offering market exclusivity and tax benefits. Increasing global awareness and advocacy from patient groups are driving demand for effective therapies and pushing for greater research investment. Furthermore, the growing pipeline of innovative treatments, particularly in gene therapy and personalized medicine, is addressing previously unmet medical needs, creating new market opportunities. The rising healthcare expenditure in emerging economies also contributes to market expansion.
Challenges in the Rare Disease Treatment Market Sector
Despite its immense growth potential, the Rare Disease Treatment Market faces considerable challenges. The inherently small patient populations for each rare disease make patient recruitment for clinical trials difficult and costly, potentially leading to delays in drug development. High research and development costs associated with complex biologics and gene therapies contribute to the significant pricing of these treatments, raising concerns about affordability and accessibility. Navigating stringent and evolving regulatory pathways for novel therapies can also be a hurdle. Furthermore, limited manufacturing capacity for specialized biologics and gene therapies can lead to supply chain disruptions. The lack of widespread diagnostic tools for many rare diseases also hinders timely diagnosis and treatment initiation.
Emerging Opportunities in Rare Disease Treatment Market
The Rare Disease Treatment Market is ripe with emerging opportunities. The rapid advancement of gene editing technologies like CRISPR-Cas9 presents a transformative avenue for developing curative therapies for a wide range of genetic disorders. The increasing adoption of real-world evidence (RWE) in clinical trials and regulatory submissions is streamlining the drug approval process and demonstrating the long-term value of rare disease treatments. Expansion into emerging markets, where healthcare infrastructure and awareness are growing, offers significant untapped potential. Collaborations between pharmaceutical companies, academic institutions, and patient advocacy groups are fostering innovative research and accelerating therapeutic development. The development of companion diagnostics to identify eligible patient populations for targeted therapies is also a key growth area.
Leading Players in the Rare Disease Treatment Market Market
- Pfizer Inc
- AstraZeneca (Alexion Pharmaceuticals Inc)
- Bayer AG
- Sanofi
- Novartis AG
- Amgen Inc
- F Hoffmann-La Roche Ltd
- Teva Pharmaceuticals
- Baxter
- Eli Lilly and Company
- Biomarin Pharmaceuticals
- Vertex Pharmaceuticals
- AbbVie Inc
- Eisai Co Ltd
- Bristol-Myers Squibb Company
Key Developments in Rare Disease Treatment Market Industry
- November 2022: ProtalixBiotherapeutics Inc. and Chiesi Global Rare Diseases resubmitted the Biologics License Application (BLA) to the United States Food and Drug Administration (FDA) for PRX-102 (pegunigalsidasealfa) for the treatment of adult patients with Fabry disease.
- October 2022: The National Institutes of Health, the US Food and Drug Administration, ten pharmaceutical companies, and five non-profit groups joined forces to speed up the development of gene therapies for the 30 million Americans who suffer from rare diseases.
Strategic Outlook for Rare Disease Treatment Market Market
The strategic outlook for the Rare Disease Treatment Market is overwhelmingly positive, driven by continuous scientific innovation and a growing global commitment to addressing rare conditions. The increasing prevalence of rare disease research, fueled by advancements in genomics and personalized medicine, will continue to unlock new therapeutic avenues. Strategic partnerships and collaborations will be crucial for navigating the complexities of drug development and market access. The market is expected to witness sustained growth, with a particular emphasis on gene and cell therapies that offer potential cures rather than just symptom management. Investments in robust manufacturing capabilities and streamlined regulatory pathways will be key to ensuring timely and widespread patient access to these life-changing treatments, solidifying its position as a critical and expanding sector within the pharmaceutical industry.
Rare Disease Treatment Market Segmentation
-
1. Drug Type
- 1.1. Biologics
- 1.2. Non-biologics
-
2. Therapeutic Area
- 2.1. Genetic Diseases
- 2.2. Neurological Diseases
- 2.3. Oncology
- 2.4. Infectious Diseases
- 2.5. Cardiovascular Diseases
- 2.6. Other Therapeutic Area
-
3. Mode of Administration
- 3.1. Oral
- 3.2. Injection
- 3.3. Other Modes of Administration
Rare Disease Treatment Market Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
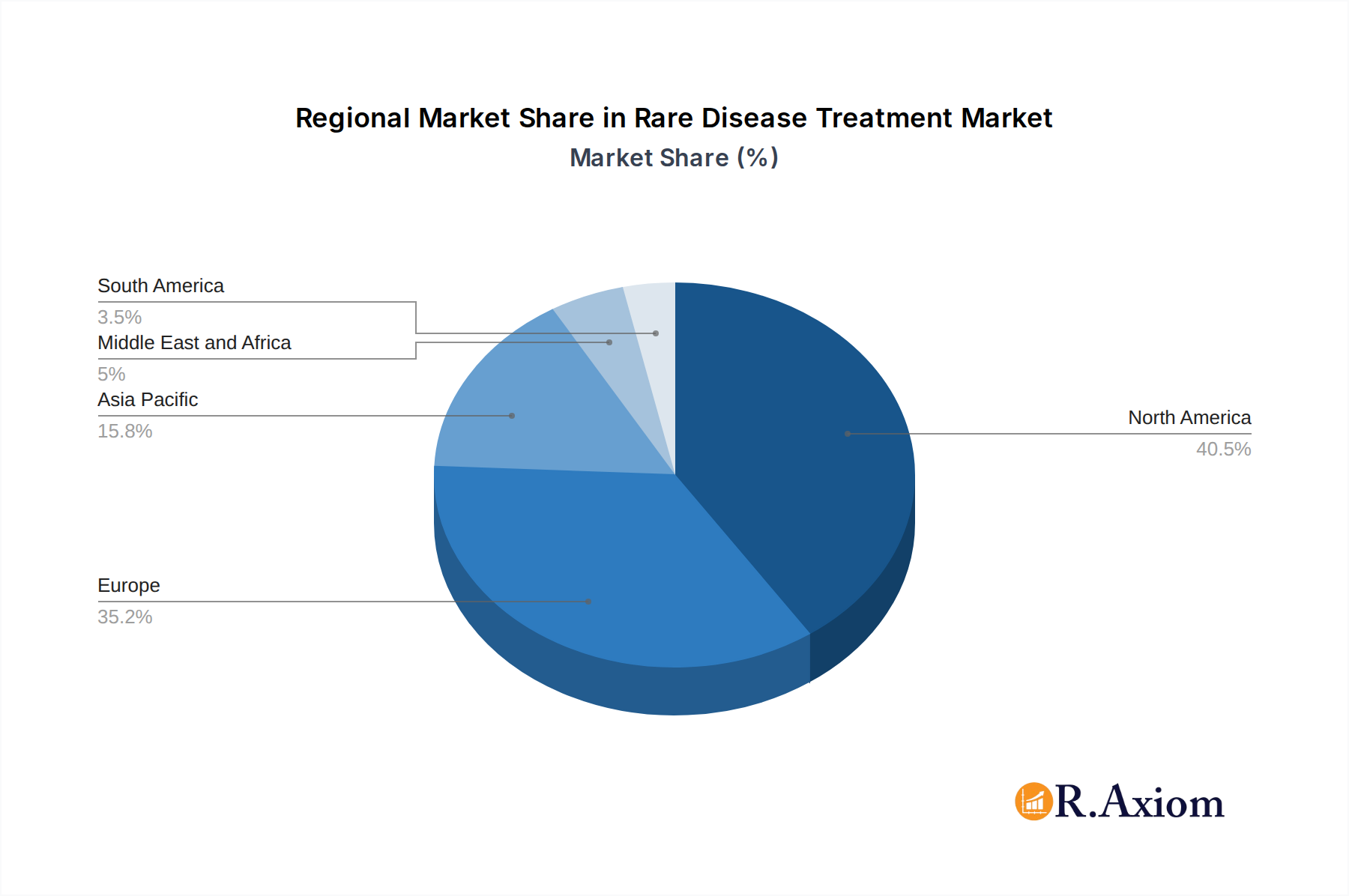
Rare Disease Treatment Market Regional Market Share

Geographic Coverage of Rare Disease Treatment Market
Rare Disease Treatment Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 11.93% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. RAX Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Drug Type
- 5.1.1. Biologics
- 5.1.2. Non-biologics
- 5.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 5.2.1. Genetic Diseases
- 5.2.2. Neurological Diseases
- 5.2.3. Oncology
- 5.2.4. Infectious Diseases
- 5.2.5. Cardiovascular Diseases
- 5.2.6. Other Therapeutic Area
- 5.3. Market Analysis, Insights and Forecast - by Mode of Administration
- 5.3.1. Oral
- 5.3.2. Injection
- 5.3.3. Other Modes of Administration
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. North America
- 5.4.2. Europe
- 5.4.3. Asia Pacific
- 5.4.4. Middle East and Africa
- 5.4.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Drug Type
- 6. Global Rare Disease Treatment Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Drug Type
- 6.1.1. Biologics
- 6.1.2. Non-biologics
- 6.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 6.2.1. Genetic Diseases
- 6.2.2. Neurological Diseases
- 6.2.3. Oncology
- 6.2.4. Infectious Diseases
- 6.2.5. Cardiovascular Diseases
- 6.2.6. Other Therapeutic Area
- 6.3. Market Analysis, Insights and Forecast - by Mode of Administration
- 6.3.1. Oral
- 6.3.2. Injection
- 6.3.3. Other Modes of Administration
- 6.1. Market Analysis, Insights and Forecast - by Drug Type
- 7. North America Rare Disease Treatment Market Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Drug Type
- 7.1.1. Biologics
- 7.1.2. Non-biologics
- 7.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 7.2.1. Genetic Diseases
- 7.2.2. Neurological Diseases
- 7.2.3. Oncology
- 7.2.4. Infectious Diseases
- 7.2.5. Cardiovascular Diseases
- 7.2.6. Other Therapeutic Area
- 7.3. Market Analysis, Insights and Forecast - by Mode of Administration
- 7.3.1. Oral
- 7.3.2. Injection
- 7.3.3. Other Modes of Administration
- 7.1. Market Analysis, Insights and Forecast - by Drug Type
- 8. Europe Rare Disease Treatment Market Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Drug Type
- 8.1.1. Biologics
- 8.1.2. Non-biologics
- 8.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 8.2.1. Genetic Diseases
- 8.2.2. Neurological Diseases
- 8.2.3. Oncology
- 8.2.4. Infectious Diseases
- 8.2.5. Cardiovascular Diseases
- 8.2.6. Other Therapeutic Area
- 8.3. Market Analysis, Insights and Forecast - by Mode of Administration
- 8.3.1. Oral
- 8.3.2. Injection
- 8.3.3. Other Modes of Administration
- 8.1. Market Analysis, Insights and Forecast - by Drug Type
- 9. Asia Pacific Rare Disease Treatment Market Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Drug Type
- 9.1.1. Biologics
- 9.1.2. Non-biologics
- 9.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 9.2.1. Genetic Diseases
- 9.2.2. Neurological Diseases
- 9.2.3. Oncology
- 9.2.4. Infectious Diseases
- 9.2.5. Cardiovascular Diseases
- 9.2.6. Other Therapeutic Area
- 9.3. Market Analysis, Insights and Forecast - by Mode of Administration
- 9.3.1. Oral
- 9.3.2. Injection
- 9.3.3. Other Modes of Administration
- 9.1. Market Analysis, Insights and Forecast - by Drug Type
- 10. Middle East and Africa Rare Disease Treatment Market Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Drug Type
- 10.1.1. Biologics
- 10.1.2. Non-biologics
- 10.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 10.2.1. Genetic Diseases
- 10.2.2. Neurological Diseases
- 10.2.3. Oncology
- 10.2.4. Infectious Diseases
- 10.2.5. Cardiovascular Diseases
- 10.2.6. Other Therapeutic Area
- 10.3. Market Analysis, Insights and Forecast - by Mode of Administration
- 10.3.1. Oral
- 10.3.2. Injection
- 10.3.3. Other Modes of Administration
- 10.1. Market Analysis, Insights and Forecast - by Drug Type
- 11. South America Rare Disease Treatment Market Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Drug Type
- 11.1.1. Biologics
- 11.1.2. Non-biologics
- 11.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 11.2.1. Genetic Diseases
- 11.2.2. Neurological Diseases
- 11.2.3. Oncology
- 11.2.4. Infectious Diseases
- 11.2.5. Cardiovascular Diseases
- 11.2.6. Other Therapeutic Area
- 11.3. Market Analysis, Insights and Forecast - by Mode of Administration
- 11.3.1. Oral
- 11.3.2. Injection
- 11.3.3. Other Modes of Administration
- 11.1. Market Analysis, Insights and Forecast - by Drug Type
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Pfizer Inc
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 AstraZeneca (Alexion Pharmaceuticals Inc )
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Bayer AG
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Sanofi
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Novartis AG
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Amgen Inc
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 F Hoffmann-La Roche Ltd
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Teva Pharmaceuticals
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Baxter
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Eli Lilly and Company
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Biomarin Pharmaceuticals
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Vertex Pharmaceuticals
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 AbbVie Inc
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.14 Eisai Co Ltd
- 12.1.14.1. Company Overview
- 12.1.14.2. Products
- 12.1.14.3. Company Financials
- 12.1.14.4. SWOT Analysis
- 12.1.15 Bristol-Myers Squibb Company
- 12.1.15.1. Company Overview
- 12.1.15.2. Products
- 12.1.15.3. Company Financials
- 12.1.15.4. SWOT Analysis
- 12.1.1 Pfizer Inc
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Rare Disease Treatment Market Revenue Breakdown (Million, %) by Region 2025 & 2033
- Figure 2: Global Rare Disease Treatment Market Volume Breakdown (K Unit, %) by Region 2025 & 2033
- Figure 3: North America Rare Disease Treatment Market Revenue (Million), by Drug Type 2025 & 2033
- Figure 4: North America Rare Disease Treatment Market Volume (K Unit), by Drug Type 2025 & 2033
- Figure 5: North America Rare Disease Treatment Market Revenue Share (%), by Drug Type 2025 & 2033
- Figure 6: North America Rare Disease Treatment Market Volume Share (%), by Drug Type 2025 & 2033
- Figure 7: North America Rare Disease Treatment Market Revenue (Million), by Therapeutic Area 2025 & 2033
- Figure 8: North America Rare Disease Treatment Market Volume (K Unit), by Therapeutic Area 2025 & 2033
- Figure 9: North America Rare Disease Treatment Market Revenue Share (%), by Therapeutic Area 2025 & 2033
- Figure 10: North America Rare Disease Treatment Market Volume Share (%), by Therapeutic Area 2025 & 2033
- Figure 11: North America Rare Disease Treatment Market Revenue (Million), by Mode of Administration 2025 & 2033
- Figure 12: North America Rare Disease Treatment Market Volume (K Unit), by Mode of Administration 2025 & 2033
- Figure 13: North America Rare Disease Treatment Market Revenue Share (%), by Mode of Administration 2025 & 2033
- Figure 14: North America Rare Disease Treatment Market Volume Share (%), by Mode of Administration 2025 & 2033
- Figure 15: North America Rare Disease Treatment Market Revenue (Million), by Country 2025 & 2033
- Figure 16: North America Rare Disease Treatment Market Volume (K Unit), by Country 2025 & 2033
- Figure 17: North America Rare Disease Treatment Market Revenue Share (%), by Country 2025 & 2033
- Figure 18: North America Rare Disease Treatment Market Volume Share (%), by Country 2025 & 2033
- Figure 19: Europe Rare Disease Treatment Market Revenue (Million), by Drug Type 2025 & 2033
- Figure 20: Europe Rare Disease Treatment Market Volume (K Unit), by Drug Type 2025 & 2033
- Figure 21: Europe Rare Disease Treatment Market Revenue Share (%), by Drug Type 2025 & 2033
- Figure 22: Europe Rare Disease Treatment Market Volume Share (%), by Drug Type 2025 & 2033
- Figure 23: Europe Rare Disease Treatment Market Revenue (Million), by Therapeutic Area 2025 & 2033
- Figure 24: Europe Rare Disease Treatment Market Volume (K Unit), by Therapeutic Area 2025 & 2033
- Figure 25: Europe Rare Disease Treatment Market Revenue Share (%), by Therapeutic Area 2025 & 2033
- Figure 26: Europe Rare Disease Treatment Market Volume Share (%), by Therapeutic Area 2025 & 2033
- Figure 27: Europe Rare Disease Treatment Market Revenue (Million), by Mode of Administration 2025 & 2033
- Figure 28: Europe Rare Disease Treatment Market Volume (K Unit), by Mode of Administration 2025 & 2033
- Figure 29: Europe Rare Disease Treatment Market Revenue Share (%), by Mode of Administration 2025 & 2033
- Figure 30: Europe Rare Disease Treatment Market Volume Share (%), by Mode of Administration 2025 & 2033
- Figure 31: Europe Rare Disease Treatment Market Revenue (Million), by Country 2025 & 2033
- Figure 32: Europe Rare Disease Treatment Market Volume (K Unit), by Country 2025 & 2033
- Figure 33: Europe Rare Disease Treatment Market Revenue Share (%), by Country 2025 & 2033
- Figure 34: Europe Rare Disease Treatment Market Volume Share (%), by Country 2025 & 2033
- Figure 35: Asia Pacific Rare Disease Treatment Market Revenue (Million), by Drug Type 2025 & 2033
- Figure 36: Asia Pacific Rare Disease Treatment Market Volume (K Unit), by Drug Type 2025 & 2033
- Figure 37: Asia Pacific Rare Disease Treatment Market Revenue Share (%), by Drug Type 2025 & 2033
- Figure 38: Asia Pacific Rare Disease Treatment Market Volume Share (%), by Drug Type 2025 & 2033
- Figure 39: Asia Pacific Rare Disease Treatment Market Revenue (Million), by Therapeutic Area 2025 & 2033
- Figure 40: Asia Pacific Rare Disease Treatment Market Volume (K Unit), by Therapeutic Area 2025 & 2033
- Figure 41: Asia Pacific Rare Disease Treatment Market Revenue Share (%), by Therapeutic Area 2025 & 2033
- Figure 42: Asia Pacific Rare Disease Treatment Market Volume Share (%), by Therapeutic Area 2025 & 2033
- Figure 43: Asia Pacific Rare Disease Treatment Market Revenue (Million), by Mode of Administration 2025 & 2033
- Figure 44: Asia Pacific Rare Disease Treatment Market Volume (K Unit), by Mode of Administration 2025 & 2033
- Figure 45: Asia Pacific Rare Disease Treatment Market Revenue Share (%), by Mode of Administration 2025 & 2033
- Figure 46: Asia Pacific Rare Disease Treatment Market Volume Share (%), by Mode of Administration 2025 & 2033
- Figure 47: Asia Pacific Rare Disease Treatment Market Revenue (Million), by Country 2025 & 2033
- Figure 48: Asia Pacific Rare Disease Treatment Market Volume (K Unit), by Country 2025 & 2033
- Figure 49: Asia Pacific Rare Disease Treatment Market Revenue Share (%), by Country 2025 & 2033
- Figure 50: Asia Pacific Rare Disease Treatment Market Volume Share (%), by Country 2025 & 2033
- Figure 51: Middle East and Africa Rare Disease Treatment Market Revenue (Million), by Drug Type 2025 & 2033
- Figure 52: Middle East and Africa Rare Disease Treatment Market Volume (K Unit), by Drug Type 2025 & 2033
- Figure 53: Middle East and Africa Rare Disease Treatment Market Revenue Share (%), by Drug Type 2025 & 2033
- Figure 54: Middle East and Africa Rare Disease Treatment Market Volume Share (%), by Drug Type 2025 & 2033
- Figure 55: Middle East and Africa Rare Disease Treatment Market Revenue (Million), by Therapeutic Area 2025 & 2033
- Figure 56: Middle East and Africa Rare Disease Treatment Market Volume (K Unit), by Therapeutic Area 2025 & 2033
- Figure 57: Middle East and Africa Rare Disease Treatment Market Revenue Share (%), by Therapeutic Area 2025 & 2033
- Figure 58: Middle East and Africa Rare Disease Treatment Market Volume Share (%), by Therapeutic Area 2025 & 2033
- Figure 59: Middle East and Africa Rare Disease Treatment Market Revenue (Million), by Mode of Administration 2025 & 2033
- Figure 60: Middle East and Africa Rare Disease Treatment Market Volume (K Unit), by Mode of Administration 2025 & 2033
- Figure 61: Middle East and Africa Rare Disease Treatment Market Revenue Share (%), by Mode of Administration 2025 & 2033
- Figure 62: Middle East and Africa Rare Disease Treatment Market Volume Share (%), by Mode of Administration 2025 & 2033
- Figure 63: Middle East and Africa Rare Disease Treatment Market Revenue (Million), by Country 2025 & 2033
- Figure 64: Middle East and Africa Rare Disease Treatment Market Volume (K Unit), by Country 2025 & 2033
- Figure 65: Middle East and Africa Rare Disease Treatment Market Revenue Share (%), by Country 2025 & 2033
- Figure 66: Middle East and Africa Rare Disease Treatment Market Volume Share (%), by Country 2025 & 2033
- Figure 67: South America Rare Disease Treatment Market Revenue (Million), by Drug Type 2025 & 2033
- Figure 68: South America Rare Disease Treatment Market Volume (K Unit), by Drug Type 2025 & 2033
- Figure 69: South America Rare Disease Treatment Market Revenue Share (%), by Drug Type 2025 & 2033
- Figure 70: South America Rare Disease Treatment Market Volume Share (%), by Drug Type 2025 & 2033
- Figure 71: South America Rare Disease Treatment Market Revenue (Million), by Therapeutic Area 2025 & 2033
- Figure 72: South America Rare Disease Treatment Market Volume (K Unit), by Therapeutic Area 2025 & 2033
- Figure 73: South America Rare Disease Treatment Market Revenue Share (%), by Therapeutic Area 2025 & 2033
- Figure 74: South America Rare Disease Treatment Market Volume Share (%), by Therapeutic Area 2025 & 2033
- Figure 75: South America Rare Disease Treatment Market Revenue (Million), by Mode of Administration 2025 & 2033
- Figure 76: South America Rare Disease Treatment Market Volume (K Unit), by Mode of Administration 2025 & 2033
- Figure 77: South America Rare Disease Treatment Market Revenue Share (%), by Mode of Administration 2025 & 2033
- Figure 78: South America Rare Disease Treatment Market Volume Share (%), by Mode of Administration 2025 & 2033
- Figure 79: South America Rare Disease Treatment Market Revenue (Million), by Country 2025 & 2033
- Figure 80: South America Rare Disease Treatment Market Volume (K Unit), by Country 2025 & 2033
- Figure 81: South America Rare Disease Treatment Market Revenue Share (%), by Country 2025 & 2033
- Figure 82: South America Rare Disease Treatment Market Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Rare Disease Treatment Market Revenue Million Forecast, by Drug Type 2020 & 2033
- Table 2: Global Rare Disease Treatment Market Volume K Unit Forecast, by Drug Type 2020 & 2033
- Table 3: Global Rare Disease Treatment Market Revenue Million Forecast, by Therapeutic Area 2020 & 2033
- Table 4: Global Rare Disease Treatment Market Volume K Unit Forecast, by Therapeutic Area 2020 & 2033
- Table 5: Global Rare Disease Treatment Market Revenue Million Forecast, by Mode of Administration 2020 & 2033
- Table 6: Global Rare Disease Treatment Market Volume K Unit Forecast, by Mode of Administration 2020 & 2033
- Table 7: Global Rare Disease Treatment Market Revenue Million Forecast, by Region 2020 & 2033
- Table 8: Global Rare Disease Treatment Market Volume K Unit Forecast, by Region 2020 & 2033
- Table 9: Global Rare Disease Treatment Market Revenue Million Forecast, by Drug Type 2020 & 2033
- Table 10: Global Rare Disease Treatment Market Volume K Unit Forecast, by Drug Type 2020 & 2033
- Table 11: Global Rare Disease Treatment Market Revenue Million Forecast, by Therapeutic Area 2020 & 2033
- Table 12: Global Rare Disease Treatment Market Volume K Unit Forecast, by Therapeutic Area 2020 & 2033
- Table 13: Global Rare Disease Treatment Market Revenue Million Forecast, by Mode of Administration 2020 & 2033
- Table 14: Global Rare Disease Treatment Market Volume K Unit Forecast, by Mode of Administration 2020 & 2033
- Table 15: Global Rare Disease Treatment Market Revenue Million Forecast, by Country 2020 & 2033
- Table 16: Global Rare Disease Treatment Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 17: United States Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 18: United States Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 19: Canada Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 20: Canada Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 21: Mexico Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 22: Mexico Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 23: Global Rare Disease Treatment Market Revenue Million Forecast, by Drug Type 2020 & 2033
- Table 24: Global Rare Disease Treatment Market Volume K Unit Forecast, by Drug Type 2020 & 2033
- Table 25: Global Rare Disease Treatment Market Revenue Million Forecast, by Therapeutic Area 2020 & 2033
- Table 26: Global Rare Disease Treatment Market Volume K Unit Forecast, by Therapeutic Area 2020 & 2033
- Table 27: Global Rare Disease Treatment Market Revenue Million Forecast, by Mode of Administration 2020 & 2033
- Table 28: Global Rare Disease Treatment Market Volume K Unit Forecast, by Mode of Administration 2020 & 2033
- Table 29: Global Rare Disease Treatment Market Revenue Million Forecast, by Country 2020 & 2033
- Table 30: Global Rare Disease Treatment Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 31: Germany Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 32: Germany Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 33: United Kingdom Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 34: United Kingdom Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 35: France Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 36: France Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 37: Italy Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 38: Italy Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 39: Spain Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 40: Spain Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 41: Rest of Europe Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 42: Rest of Europe Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 43: Global Rare Disease Treatment Market Revenue Million Forecast, by Drug Type 2020 & 2033
- Table 44: Global Rare Disease Treatment Market Volume K Unit Forecast, by Drug Type 2020 & 2033
- Table 45: Global Rare Disease Treatment Market Revenue Million Forecast, by Therapeutic Area 2020 & 2033
- Table 46: Global Rare Disease Treatment Market Volume K Unit Forecast, by Therapeutic Area 2020 & 2033
- Table 47: Global Rare Disease Treatment Market Revenue Million Forecast, by Mode of Administration 2020 & 2033
- Table 48: Global Rare Disease Treatment Market Volume K Unit Forecast, by Mode of Administration 2020 & 2033
- Table 49: Global Rare Disease Treatment Market Revenue Million Forecast, by Country 2020 & 2033
- Table 50: Global Rare Disease Treatment Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 51: China Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 52: China Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 53: Japan Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 54: Japan Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 55: India Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 56: India Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 57: Australia Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 58: Australia Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 59: South Korea Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 60: South Korea Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 61: Rest of Asia Pacific Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 62: Rest of Asia Pacific Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 63: Global Rare Disease Treatment Market Revenue Million Forecast, by Drug Type 2020 & 2033
- Table 64: Global Rare Disease Treatment Market Volume K Unit Forecast, by Drug Type 2020 & 2033
- Table 65: Global Rare Disease Treatment Market Revenue Million Forecast, by Therapeutic Area 2020 & 2033
- Table 66: Global Rare Disease Treatment Market Volume K Unit Forecast, by Therapeutic Area 2020 & 2033
- Table 67: Global Rare Disease Treatment Market Revenue Million Forecast, by Mode of Administration 2020 & 2033
- Table 68: Global Rare Disease Treatment Market Volume K Unit Forecast, by Mode of Administration 2020 & 2033
- Table 69: Global Rare Disease Treatment Market Revenue Million Forecast, by Country 2020 & 2033
- Table 70: Global Rare Disease Treatment Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 71: GCC Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 72: GCC Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 73: South Africa Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 74: South Africa Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 75: Rest of Middle East and Africa Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 76: Rest of Middle East and Africa Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 77: Global Rare Disease Treatment Market Revenue Million Forecast, by Drug Type 2020 & 2033
- Table 78: Global Rare Disease Treatment Market Volume K Unit Forecast, by Drug Type 2020 & 2033
- Table 79: Global Rare Disease Treatment Market Revenue Million Forecast, by Therapeutic Area 2020 & 2033
- Table 80: Global Rare Disease Treatment Market Volume K Unit Forecast, by Therapeutic Area 2020 & 2033
- Table 81: Global Rare Disease Treatment Market Revenue Million Forecast, by Mode of Administration 2020 & 2033
- Table 82: Global Rare Disease Treatment Market Volume K Unit Forecast, by Mode of Administration 2020 & 2033
- Table 83: Global Rare Disease Treatment Market Revenue Million Forecast, by Country 2020 & 2033
- Table 84: Global Rare Disease Treatment Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 85: Brazil Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 86: Brazil Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 87: Argentina Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 88: Argentina Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 89: Rest of South America Rare Disease Treatment Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 90: Rest of South America Rare Disease Treatment Market Volume (K Unit) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Rare Disease Treatment Market?
The projected CAGR is approximately 11.93%.
2. Which companies are prominent players in the Rare Disease Treatment Market?
Key companies in the market include Pfizer Inc, AstraZeneca (Alexion Pharmaceuticals Inc ), Bayer AG, Sanofi, Novartis AG, Amgen Inc, F Hoffmann-La Roche Ltd, Teva Pharmaceuticals, Baxter, Eli Lilly and Company, Biomarin Pharmaceuticals, Vertex Pharmaceuticals, AbbVie Inc, Eisai Co Ltd, Bristol-Myers Squibb Company.
3. What are the main segments of the Rare Disease Treatment Market?
The market segments include Drug Type, Therapeutic Area, Mode of Administration.
4. Can you provide details about the market size?
The market size is estimated to be USD 216.65 Million as of 2022.
5. What are some drivers contributing to market growth?
Increase in the Number of Rare Disease Cases; Rising R&D Activities for Novel Therapeutics and Drugs and Increase in the Number of New Drug Launches and Favorable Government Policies.
6. What are the notable trends driving market growth?
The Biologics Segment is Expected to Hold a Major Market Share in the Rare Disease Treatment Market.
7. Are there any restraints impacting market growth?
Lack of Awareness Regarding Rare Disease Treatment; High Cost of Treatment.
8. Can you provide examples of recent developments in the market?
November 2022: ProtalixBiotherapeutics Inc. and Chiesi Global Rare Diseases resubmitted the Biologics License Application (BLA) to the United States Food and Drug Administration (FDA) for PRX-102 (pegunigalsidasealfa) for the treatment of adult patients with Fabry disease.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Rare Disease Treatment Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Rare Disease Treatment Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Rare Disease Treatment Market?
To stay informed about further developments, trends, and reports in the Rare Disease Treatment Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
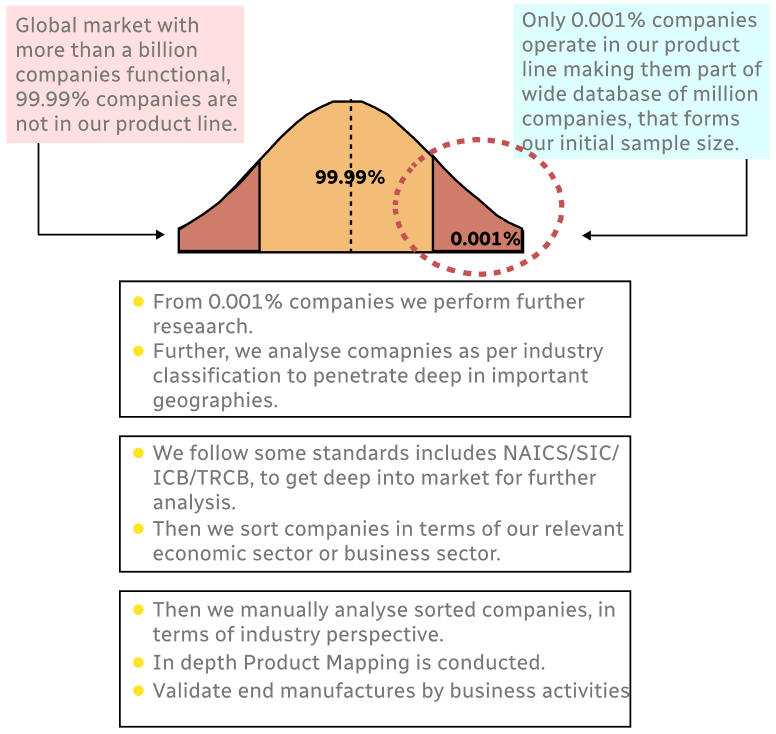
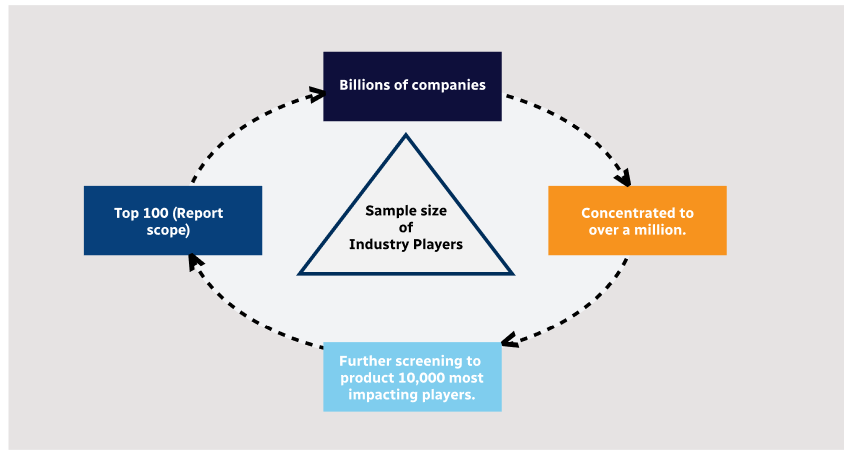
Step 1 - Identification of Relevant Samples Size from Population Database
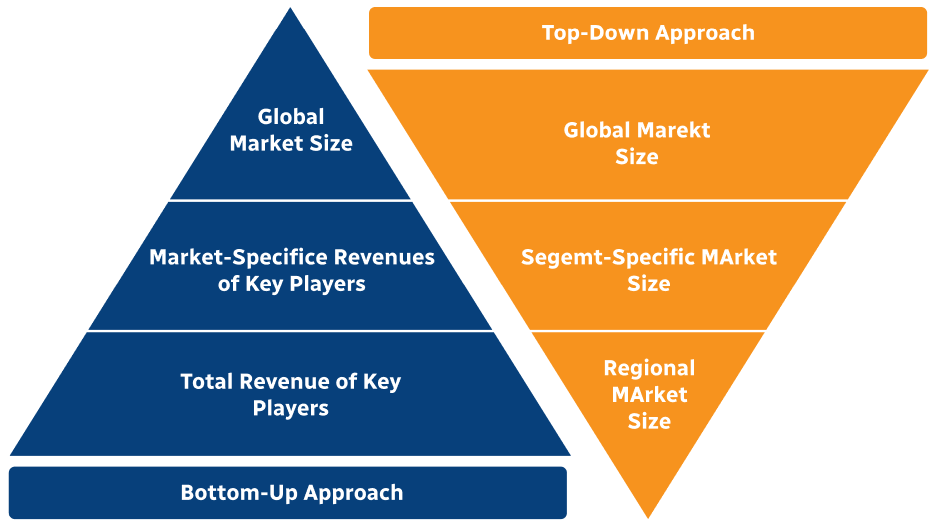
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence