Key Insights
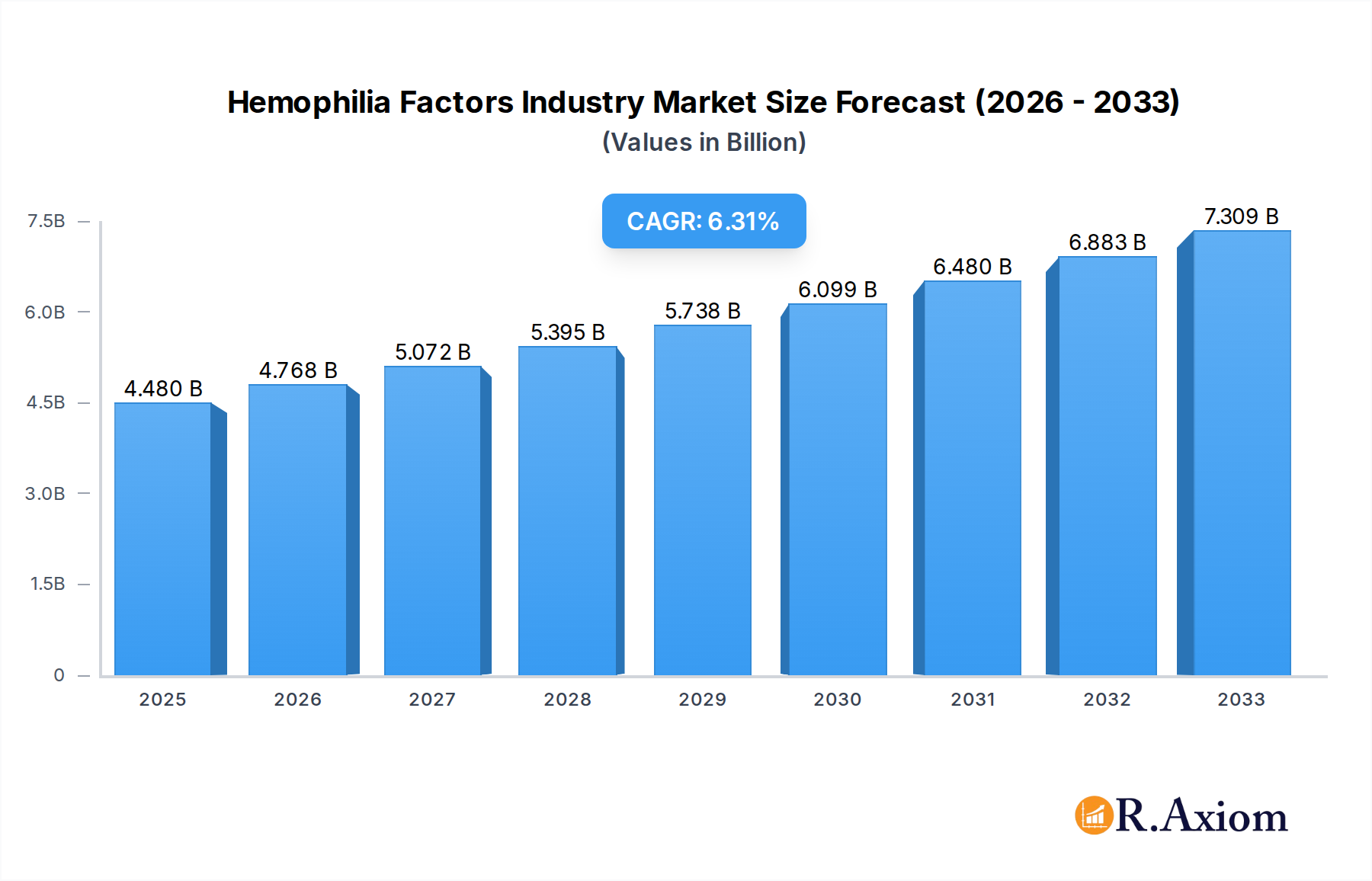
The global Hemophilia Factors market is poised for robust growth, projected to reach $4.48 billion in 2025, with a compelling Compound Annual Growth Rate (CAGR) of 6.4% through 2033. This expansion is primarily fueled by advancements in treatment therapies, including a greater adoption of factor concentrates, fresh frozen plasma (FFP), and cryoprecipitate. The increasing prevalence of hemophilia globally, coupled with rising healthcare expenditure and improved diagnostic capabilities, are significant drivers. Furthermore, a growing awareness of hemophilia management and the availability of novel therapeutic approaches are contributing to this positive market trajectory. The market is witnessing substantial investment in research and development, leading to the introduction of more effective and targeted treatments, which are crucial for improving the quality of life for individuals with hemophilia.
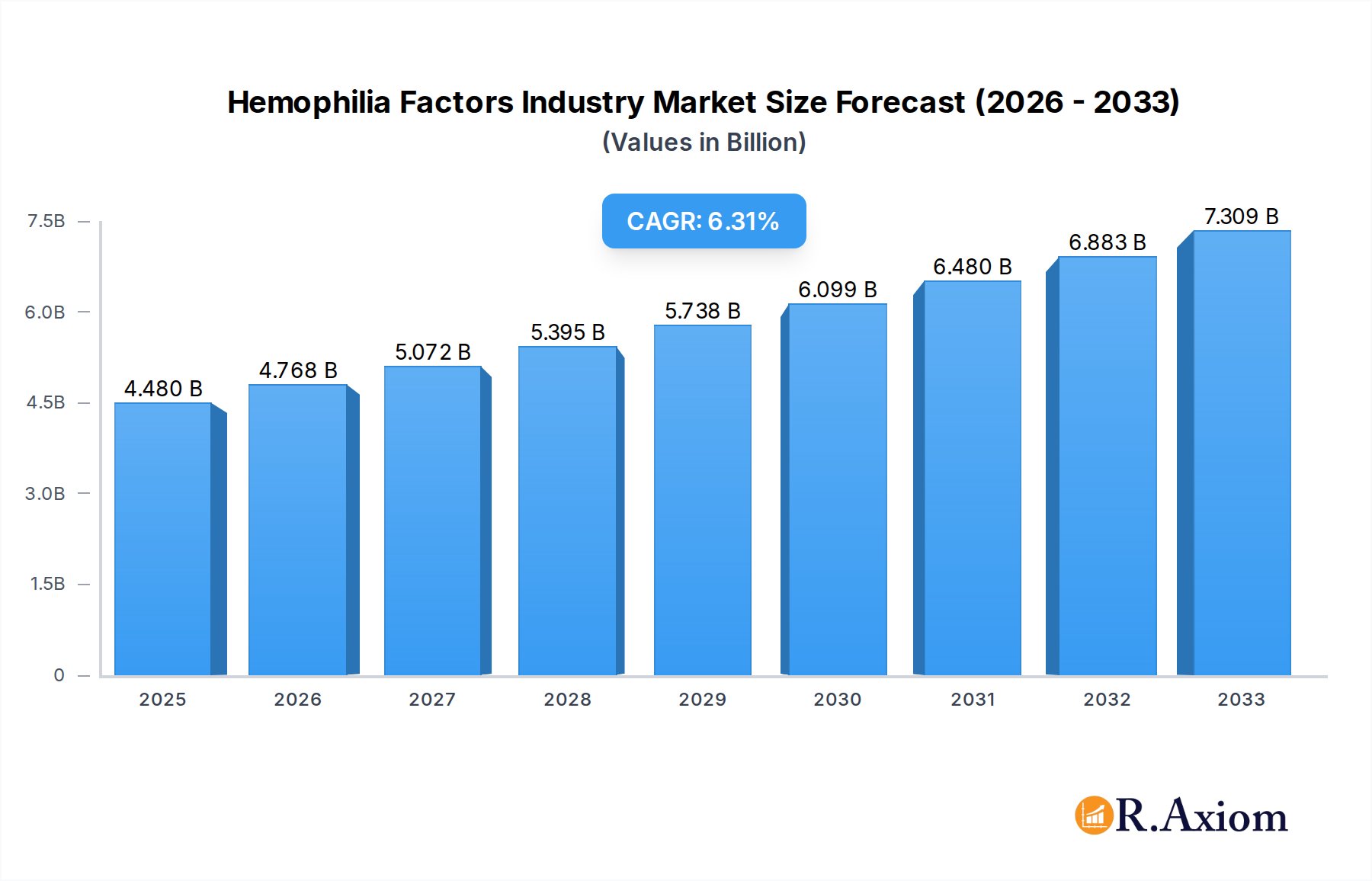
Hemophilia Factors Industry Market Size (In Billion)

The competitive landscape of the Hemophilia Factors market is characterized by the presence of key global players such as Bayer AG, Takeda Pharmaceutical Co Ltd, Novo Nordisk A/S, and Pfizer Inc, among others. These companies are actively engaged in strategic collaborations, mergers, and acquisitions to expand their product portfolios and geographical reach. Innovations in gene therapy and recombinant factor therapies are emerging trends expected to significantly influence market dynamics in the coming years. However, challenges such as the high cost of treatment, limited access to specialized care in developing regions, and potential side effects associated with certain therapies act as restraints. Despite these challenges, the market is expected to witness sustained demand, particularly in North America and Europe, with the Asia Pacific region demonstrating substantial growth potential due to improving healthcare infrastructure and increasing patient populations.
Hemophilia Factors Industry Company Market Share

This in-depth report provides an exhaustive analysis of the global Hemophilia Factors Industry, covering market dynamics, trends, segmentation, competitive landscape, and future outlook. With a study period spanning from 2019 to 2033, a base year of 2025, and a forecast period of 2025–2033, this report equips industry stakeholders with critical data and actionable insights to navigate the evolving hemophilia treatment market. The historical period analyzed is 2019–2024. Our comprehensive research delves into key segments such as Factor Concentrate, Fresh Frozen Plasma (FFP), Cryoprecipitate, and Others, alongside an evaluation of dominant players.
Hemophilia Factors Industry Market Concentration & Innovation
The Hemophilia Factors Industry exhibits a moderate to high market concentration, with a few key players holding significant market share. Innovation is a critical driver, fueled by advancements in gene therapy, extended half-life factor concentrates, and novel prophylactic treatments. The global market size for hemophilia factors is projected to reach over $20 billion by 2033, with an estimated Compound Annual Growth Rate (CAGR) of approximately 6.5% during the forecast period. Regulatory frameworks, such as those enforced by the FDA and EMA, play a pivotal role in shaping market access and product approvals, fostering a competitive yet regulated environment. Product substitutes, while emerging, are currently limited in widespread adoption, with factor replacement therapy remaining the cornerstone of treatment. End-user trends indicate a growing demand for less frequent dosing and improved quality of life for patients. Mergers and acquisitions (M&A) activities are strategic for expanding portfolios and geographical reach. Notable M&A deal values in the past have ranged from $500 million to over $2 billion, indicating significant consolidation potential.
- Market Share Drivers: Technological innovation, patent expirations, clinical trial successes, and strategic partnerships.
- Innovation Focus Areas: Gene therapy, biosimil development, personalized medicine, and improved delivery systems.
- Regulatory Landscape: Strict approval processes, post-market surveillance, and evolving guidelines for orphan drugs.
- Product Substitutes: While gene therapy is a promising long-term substitute, it is still in early stages of adoption.
- End-User Preferences: Increased demand for convenience, reduced treatment burden, and improved safety profiles.
- M&A Activity: Driven by portfolio expansion, market access, and R&D pipeline enhancement.
Hemophilia Factors Industry Industry Trends & Insights
The Hemophilia Factors Industry is on a robust growth trajectory, driven by a confluence of factors including increasing hemophilia diagnoses, enhanced diagnostic capabilities, and a growing understanding of the disease. The market is projected to expand significantly, with an estimated market size of $18 billion in 2025, escalating to over $20 billion by 2033, reflecting a healthy CAGR of approximately 6.5%. Technological disruptions are at the forefront, with the development of extended half-life factor concentrates (e.g., Adynovate) revolutionizing treatment paradigms by reducing the frequency of infusions, thereby improving patient adherence and quality of life. Gene therapy also represents a paradigm shift, offering the potential for a one-time curative treatment, though it is still in its nascent stages of widespread clinical application. Consumer preferences are increasingly shifting towards less invasive and more convenient treatment options. This includes a demand for home-infusion therapies and prophylactic regimens that minimize spontaneous bleeds. Competitive dynamics are characterized by intense R&D efforts, strategic alliances, and a focus on securing intellectual property. Pharmaceutical giants are heavily investing in rare disease research, aiming to capture a larger share of this lucrative market. The penetration of advanced therapies is steadily increasing, particularly in developed economies, while emerging markets present significant untapped potential. Challenges include high treatment costs, reimbursement complexities, and the need for specialized medical infrastructure. However, the development of biosimil factor concentrates is expected to offer more affordable treatment options, potentially increasing market penetration in cost-sensitive regions.
- Market Growth Drivers: Rising prevalence of hemophilia, improved diagnostics, increasing healthcare expenditure in emerging economies, and favorable reimbursement policies for rare diseases.
- Technological Disruptions: Gene therapy, extended half-life factor concentrates, bispecific antibodies, and novel drug delivery systems.
- Consumer Preferences: Demand for convenient administration, reduced infusion frequency, prophylactic treatment options, and improved patient outcomes.
- Competitive Dynamics: Intense R&D focus, strategic collaborations, patent cliffs, and market entry of biosimil products.
- Market Penetration: Driven by new product launches, increased awareness, and efforts to improve access in underserved populations.
Dominant Markets & Segments in Hemophilia Factors Industry
The Factor Concentrate segment is the dominant force within the Hemophilia Factors Industry, accounting for an estimated 85% of the total market value in 2025. This dominance is attributed to its established efficacy, widespread availability, and the continuous innovation in recombinant factor production and extended half-life formulations. North America, particularly the United States, represents the leading regional market due to high healthcare spending, advanced diagnostic and treatment infrastructure, and strong government support for rare disease research and patient advocacy. The market size for the Hemophilia Factors Industry is projected to be approximately $18 billion in 2025, with the Factor Concentrate segment alone contributing around $15.3 billion.
- Factor Concentrate:
- Key Drivers: Superior efficacy in managing bleeding episodes, availability of both on-demand and prophylactic treatments, continuous innovation leading to extended half-life products, and a well-established manufacturing base.
- Dominance Analysis: This segment benefits from decades of research and development, leading to highly purified and effective recombinant factor VIII and factor IX concentrates. The introduction of extended half-life formulations has further cemented its leadership by addressing the patient need for less frequent dosing.
- Fresh Frozen Plasma (FFP):
- Key Drivers: Availability as an alternative in resource-limited settings, its role in managing complex coagulation factor deficiencies, and its use in specific surgical procedures.
- Dominance Analysis: While FFP is a crucial component of hemophilia management, its use is generally secondary to factor concentrates due to its limited shelf-life, potential for transfusion-related adverse events, and variability in factor content. Its market share is estimated to be around 10% of the total market.
- Cryoprecipitate:
- Key Drivers: Primarily used for managing von Willebrand disease and fibrinogen deficiency, and as an adjunct treatment in some hemophilia cases.
- Dominance Analysis: Cryoprecipitate is a more specialized product with a smaller market share, estimated at 3%. Its application is often specific to certain factor deficiencies and is not a primary treatment for hemophilia A or B.
- Others:
- Key Drivers: Includes emerging therapies like gene therapy and bispecific antibodies, which are gaining traction but are not yet widely adopted.
- Dominance Analysis: This segment, though currently small at approximately 2%, holds significant future growth potential as gene therapies become more accessible and proven effective.
Hemophilia Factors Industry Product Developments
The Hemophilia Factors Industry is characterized by rapid product innovation aimed at improving patient outcomes and treatment convenience. A significant development includes extended half-life (EHL) factor concentrates, such as Takeda's Adynovate, which significantly reduce the frequency of infusions for hemophilia A patients. Furthermore, the United States Food and Drug Administration's (FDA) breakthrough therapy designation for efanesoctocog alfa in June 2022 highlights the potential of novel factor VIII therapies to offer superior bleed prevention compared to existing prophylactic treatments. These advancements focus on enhancing therapeutic efficacy, minimizing treatment burden, and exploring novel modalities like gene therapy, positioning the industry for substantial growth and improved patient care.
Report Scope & Segmentation Analysis
This report meticulously analyzes the Hemophilia Factors Industry across its key treatment segments. The segmentation includes Factor Concentrate, Fresh Frozen Plasma (FFP), Cryoprecipitate, and Others.
- Factor Concentrate: This segment is projected to maintain its market leadership, with an estimated market size of over $15.3 billion in 2025 and a projected growth to over $17 billion by 2033. Competitive dynamics are driven by innovation in recombinant and extended half-life formulations.
- Fresh Frozen Plasma (FFP): Expected to hold a stable market share of around 10%, with a market size of approximately $1.8 billion in 2025, growing moderately to $2 billion by 2033. Its use remains critical in specific clinical scenarios and resource-limited settings.
- Cryoprecipitate: This segment is anticipated to contribute approximately 3% to the market, valued at around $540 million in 2025 and reaching $600 million by 2033. Its application remains niche for specific factor deficiencies.
- Others: This segment, encompassing emerging therapies like gene therapy, is expected to exhibit the highest growth rate. Currently valued at around $360 million in 2025, it is projected to expand rapidly to over $1 billion by 2033, driven by advancements in gene editing and delivery technologies.
Key Drivers of Hemophilia Factors Industry Growth
The Hemophilia Factors Industry is propelled by several key drivers. Advancements in biotechnology have led to the development of highly effective recombinant factor concentrates and extended half-life therapies, significantly improving patient management and quality of life. Increased global awareness and improved diagnostic capabilities are leading to earlier and more accurate identification of hemophilia cases. Favorable regulatory pathways for orphan drugs and incentives for rare disease research encourage pharmaceutical companies to invest in this sector. Furthermore, rising healthcare expenditure, particularly in emerging economies, and expanding access to specialized treatment centers contribute to market growth.
Challenges in the Hemophilia Factors Industry Sector
Despite significant progress, the Hemophilia Factors Industry faces several challenges. The high cost of hemophilia treatments, especially novel therapies, poses a significant barrier to access, particularly in low- and middle-income countries. Reimbursement complexities and the need for stringent clinical evidence to secure coverage can also slow down market adoption. Supply chain vulnerabilities and the reliance on specialized manufacturing processes for biological products can lead to potential disruptions. Moreover, the development and approval of biosimilar factor concentrates introduce competitive pressures, necessitating continuous innovation from originator companies.
Emerging Opportunities in Hemophilia Factors Industry
Emerging opportunities in the Hemophilia Factors Industry are primarily centered around the transformative potential of gene therapy, which promises a functional cure for hemophilia. The development of bispecific antibodies offers an alternative treatment option for patients who have developed inhibitors to factor VIII. Expansion into emerging markets presents a substantial growth avenue, as awareness and diagnostic capabilities improve. Furthermore, the focus on personalized medicine and the development of targeted therapies tailored to individual patient profiles are creating new avenues for innovation and market penetration.
Leading Players in the Hemophilia Factors Industry Market
- Bayer AG
- Takeda Pharmaceutical Co Ltd
- Bio Products Laboratory Ltd
- Novo Nordisk A/S
- Biogen Inc
- Baxter International Inc
- CSL Behring
- Pfizer Inc
Key Developments in Hemophilia Factors Industry Industry
- June 2022: The United States Food and Drug Administration granted breakthrough therapy designation to 'efanesoctocog alfa' for hemophilia A. Efanesoctocog alfa is the first factor VIII therapy to be awarded Breakthrough Therapy designation by the FDA. This designation is based on XTEND-1 Phase 3 study data demonstrating clinically meaningful prevention of bleeds and superiority in the prevention of bleeding episodes compared to prior prophylaxis factor treatment.
- May 2022: Takeda Pharmaceutical Co Ltd announced the expansion of its rare diseases portfolio in India with the launch of Adynovate. Adynovate is an innovative extended half-life recombinant Factor VIII (rFVIII) treatment, using established technology for hemophilia A patients.
Strategic Outlook for Hemophilia Factors Industry Market
The Hemophilia Factors Industry is poised for continued robust growth, driven by a strong pipeline of innovative therapies and increasing global demand. The strategic focus will remain on developing and commercializing gene therapies, extended half-life factor concentrates, and other novel treatment modalities that offer improved efficacy, convenience, and patient outcomes. Expansion into emerging markets, coupled with efforts to address cost and accessibility challenges, will be crucial for unlocking future growth potential. Collaborations between pharmaceutical companies, research institutions, and patient advocacy groups will be instrumental in accelerating research and development and ensuring wider patient access to life-changing treatments.
Hemophilia Factors Industry Segmentation
-
1. Treatment
- 1.1. Factor Concentrate
- 1.2. Fresh Frozen Plasma (FFP)
- 1.3. Cryoprecipitate
- 1.4. Others
Hemophilia Factors Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
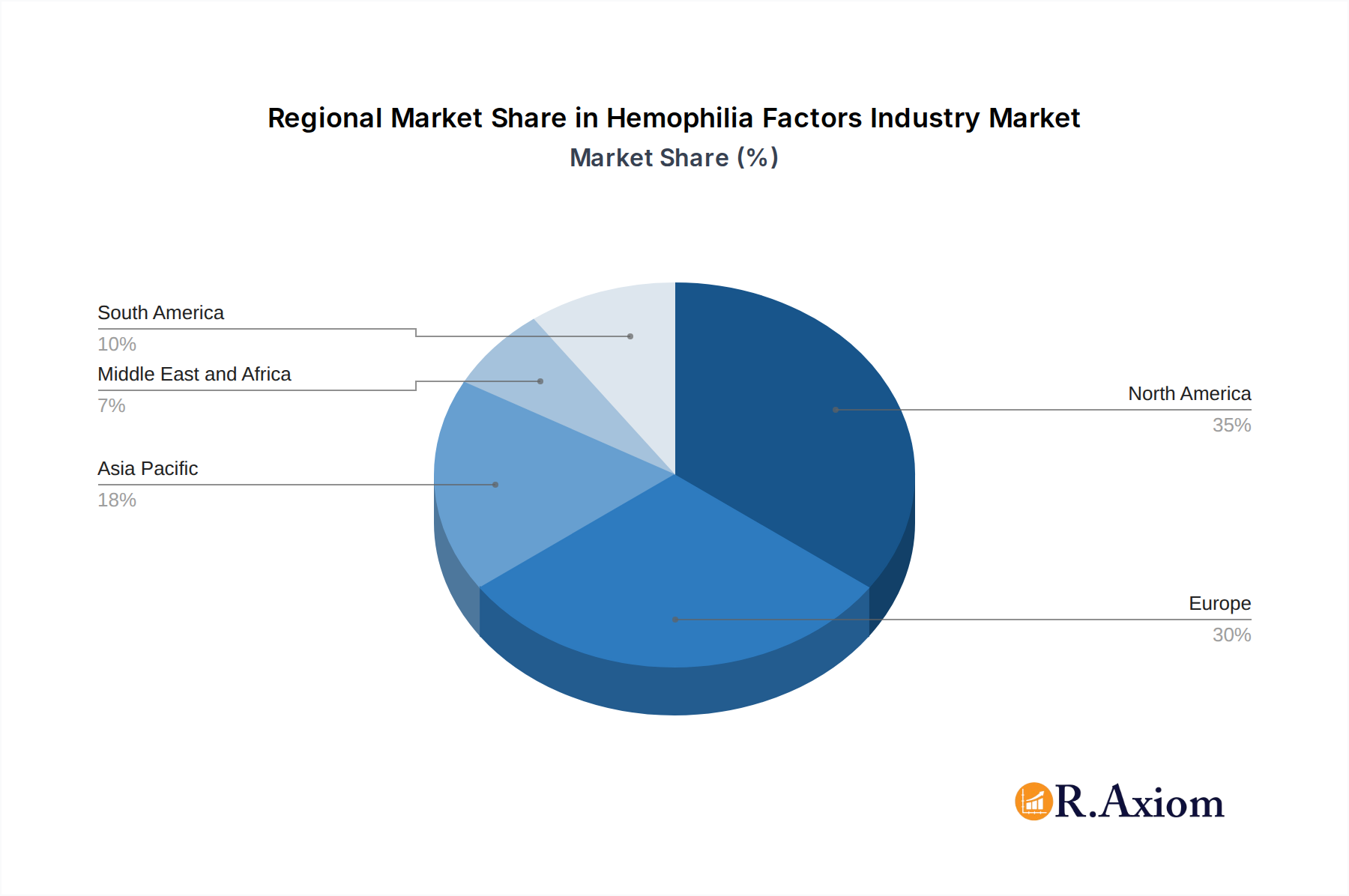
Hemophilia Factors Industry Regional Market Share

Geographic Coverage of Hemophilia Factors Industry
Hemophilia Factors Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.4% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. RAX Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Treatment
- 5.1.1. Factor Concentrate
- 5.1.2. Fresh Frozen Plasma (FFP)
- 5.1.3. Cryoprecipitate
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Region
- 5.2.1. North America
- 5.2.2. Europe
- 5.2.3. Asia Pacific
- 5.2.4. Middle East and Africa
- 5.2.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Treatment
- 6. Global Hemophilia Factors Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Treatment
- 6.1.1. Factor Concentrate
- 6.1.2. Fresh Frozen Plasma (FFP)
- 6.1.3. Cryoprecipitate
- 6.1.4. Others
- 6.1. Market Analysis, Insights and Forecast - by Treatment
- 7. North America Hemophilia Factors Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Treatment
- 7.1.1. Factor Concentrate
- 7.1.2. Fresh Frozen Plasma (FFP)
- 7.1.3. Cryoprecipitate
- 7.1.4. Others
- 7.1. Market Analysis, Insights and Forecast - by Treatment
- 8. Europe Hemophilia Factors Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Treatment
- 8.1.1. Factor Concentrate
- 8.1.2. Fresh Frozen Plasma (FFP)
- 8.1.3. Cryoprecipitate
- 8.1.4. Others
- 8.1. Market Analysis, Insights and Forecast - by Treatment
- 9. Asia Pacific Hemophilia Factors Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Treatment
- 9.1.1. Factor Concentrate
- 9.1.2. Fresh Frozen Plasma (FFP)
- 9.1.3. Cryoprecipitate
- 9.1.4. Others
- 9.1. Market Analysis, Insights and Forecast - by Treatment
- 10. Middle East and Africa Hemophilia Factors Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Treatment
- 10.1.1. Factor Concentrate
- 10.1.2. Fresh Frozen Plasma (FFP)
- 10.1.3. Cryoprecipitate
- 10.1.4. Others
- 10.1. Market Analysis, Insights and Forecast - by Treatment
- 11. South America Hemophilia Factors Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Treatment
- 11.1.1. Factor Concentrate
- 11.1.2. Fresh Frozen Plasma (FFP)
- 11.1.3. Cryoprecipitate
- 11.1.4. Others
- 11.1. Market Analysis, Insights and Forecast - by Treatment
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Bayer AG
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Takeda Pharmaceutical Co Ltd *List Not Exhaustive
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Bio Products Laboratory Ltd
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Novo Nordisk A/S
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Biogen Inc
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Baxter International Inc
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 CSL Behring
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Pfizer Inc
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.1 Bayer AG
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Hemophilia Factors Industry Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: North America Hemophilia Factors Industry Revenue (billion), by Treatment 2025 & 2033
- Figure 3: North America Hemophilia Factors Industry Revenue Share (%), by Treatment 2025 & 2033
- Figure 4: North America Hemophilia Factors Industry Revenue (billion), by Country 2025 & 2033
- Figure 5: North America Hemophilia Factors Industry Revenue Share (%), by Country 2025 & 2033
- Figure 6: Europe Hemophilia Factors Industry Revenue (billion), by Treatment 2025 & 2033
- Figure 7: Europe Hemophilia Factors Industry Revenue Share (%), by Treatment 2025 & 2033
- Figure 8: Europe Hemophilia Factors Industry Revenue (billion), by Country 2025 & 2033
- Figure 9: Europe Hemophilia Factors Industry Revenue Share (%), by Country 2025 & 2033
- Figure 10: Asia Pacific Hemophilia Factors Industry Revenue (billion), by Treatment 2025 & 2033
- Figure 11: Asia Pacific Hemophilia Factors Industry Revenue Share (%), by Treatment 2025 & 2033
- Figure 12: Asia Pacific Hemophilia Factors Industry Revenue (billion), by Country 2025 & 2033
- Figure 13: Asia Pacific Hemophilia Factors Industry Revenue Share (%), by Country 2025 & 2033
- Figure 14: Middle East and Africa Hemophilia Factors Industry Revenue (billion), by Treatment 2025 & 2033
- Figure 15: Middle East and Africa Hemophilia Factors Industry Revenue Share (%), by Treatment 2025 & 2033
- Figure 16: Middle East and Africa Hemophilia Factors Industry Revenue (billion), by Country 2025 & 2033
- Figure 17: Middle East and Africa Hemophilia Factors Industry Revenue Share (%), by Country 2025 & 2033
- Figure 18: South America Hemophilia Factors Industry Revenue (billion), by Treatment 2025 & 2033
- Figure 19: South America Hemophilia Factors Industry Revenue Share (%), by Treatment 2025 & 2033
- Figure 20: South America Hemophilia Factors Industry Revenue (billion), by Country 2025 & 2033
- Figure 21: South America Hemophilia Factors Industry Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Hemophilia Factors Industry Revenue billion Forecast, by Treatment 2020 & 2033
- Table 2: Global Hemophilia Factors Industry Revenue billion Forecast, by Region 2020 & 2033
- Table 3: Global Hemophilia Factors Industry Revenue billion Forecast, by Treatment 2020 & 2033
- Table 4: Global Hemophilia Factors Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 5: United States Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 6: Canada Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 7: Mexico Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 8: Global Hemophilia Factors Industry Revenue billion Forecast, by Treatment 2020 & 2033
- Table 9: Global Hemophilia Factors Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 10: Germany Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 11: United Kingdom Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 12: France Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 13: Italy Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: Spain Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 15: Rest of Europe Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Global Hemophilia Factors Industry Revenue billion Forecast, by Treatment 2020 & 2033
- Table 17: Global Hemophilia Factors Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 18: China Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 19: Japan Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 20: India Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 21: Australia Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 22: South Korea Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 23: Rest of Asia Pacific Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 24: Global Hemophilia Factors Industry Revenue billion Forecast, by Treatment 2020 & 2033
- Table 25: Global Hemophilia Factors Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 26: GCC Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 27: South Africa Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Rest of Middle East and Africa Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 29: Global Hemophilia Factors Industry Revenue billion Forecast, by Treatment 2020 & 2033
- Table 30: Global Hemophilia Factors Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 31: Brazil Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: Argentina Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 33: Rest of South America Hemophilia Factors Industry Revenue (billion) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Hemophilia Factors Industry?
The projected CAGR is approximately 6.4%.
2. Which companies are prominent players in the Hemophilia Factors Industry?
Key companies in the market include Bayer AG, Takeda Pharmaceutical Co Ltd *List Not Exhaustive, Bio Products Laboratory Ltd, Novo Nordisk A/S, Biogen Inc, Baxter International Inc, CSL Behring, Pfizer Inc.
3. What are the main segments of the Hemophilia Factors Industry?
The market segments include Treatment.
4. Can you provide details about the market size?
The market size is estimated to be USD 4.48 billion as of 2022.
5. What are some drivers contributing to market growth?
Rising Adoption and Approval of New Treatment Techniques; Growing Number of Government Initiatives and Funding.
6. What are the notable trends driving market growth?
Fresh Frozen Plasma (FFP) is Expected to Hold Significant Market Share in the Treatment Segment.
7. Are there any restraints impacting market growth?
High Cost of Treatment.
8. Can you provide examples of recent developments in the market?
In June 2022, the United States Food and Drug Administration granted breakthrough therapy designation to 'efanesoctocog alfa' for hemophilia A.Efanesoctocog alfa is the first factor VIII therapy to be awarded Breakthrough Therapy designation by the FDA. This designation is based on XTEND-1 Phase 3 study data demonstrating clinically meaningful prevention of bleeds and superiority in the prevention of bleeding episodes compared to prior prophylaxis factor treatment.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Hemophilia Factors Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Hemophilia Factors Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Hemophilia Factors Industry?
To stay informed about further developments, trends, and reports in the Hemophilia Factors Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
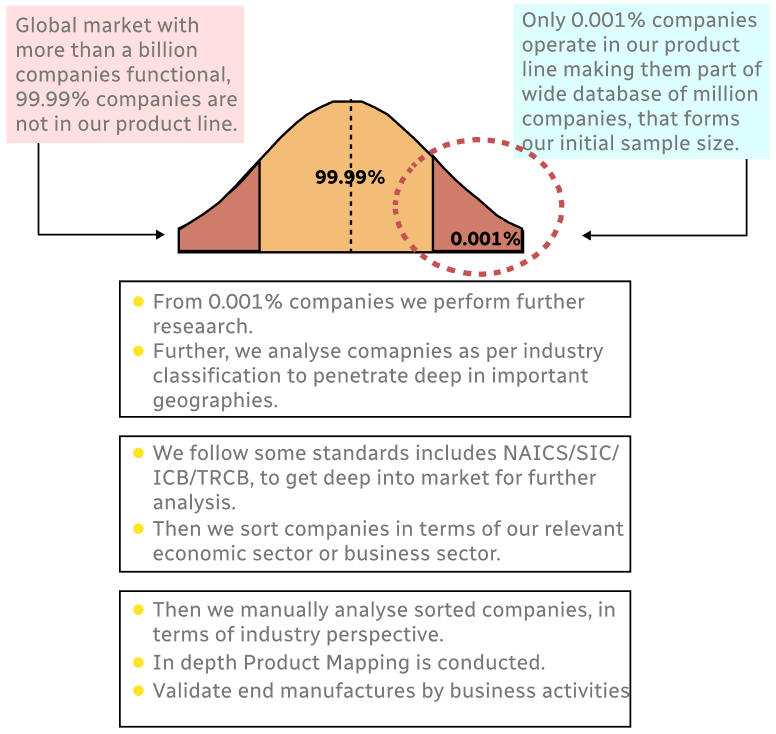
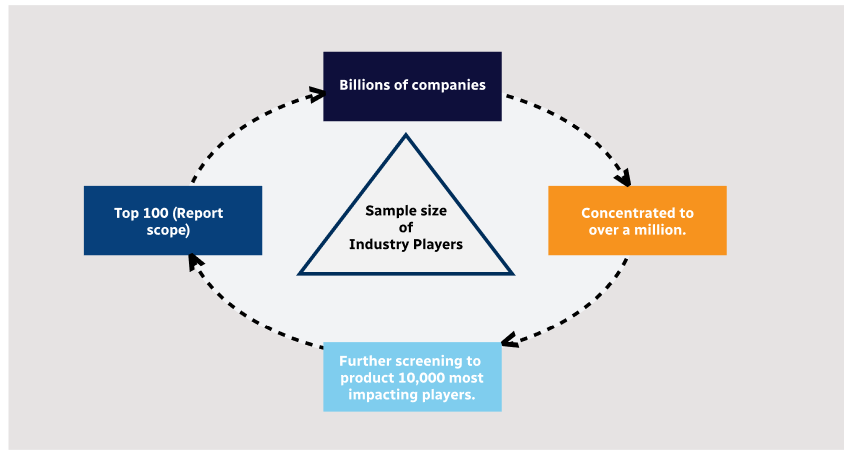
Step 1 - Identification of Relevant Samples Size from Population Database
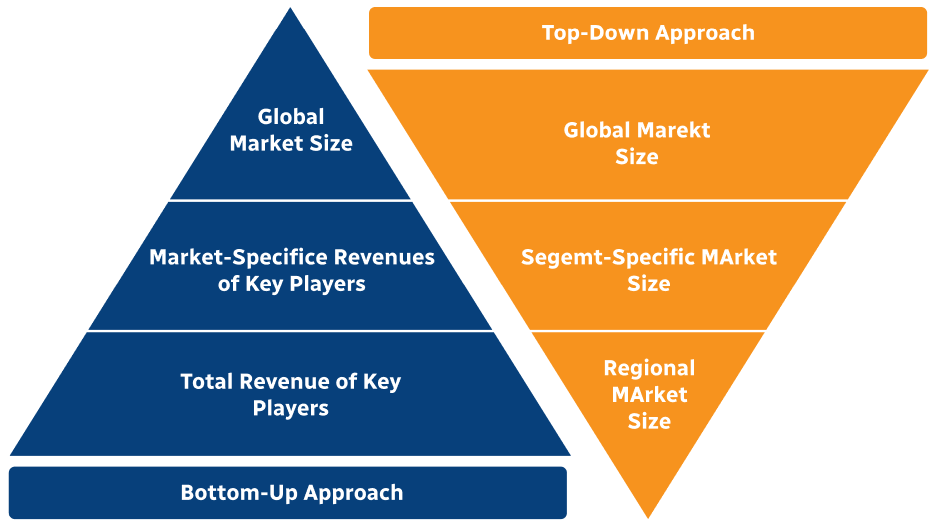
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence