Key Insights
The Lysosomal Storage Disease (LSD) Treatment market is poised for significant expansion, projected to reach an estimated USD 10 billion in 2025, with a robust Compound Annual Growth Rate (CAGR) of 6.9% during the forecast period of 2025-2033. This growth is primarily fueled by advancements in therapeutic approaches such as Enzyme Replacement Therapy (ERT) and Substrate Reduction Therapy (SRT), which are becoming increasingly effective and accessible. The growing understanding of rare genetic disorders, coupled with enhanced diagnostic capabilities and a rising prevalence of lysosomal storage diseases, particularly in developed regions, are key drivers. Furthermore, increased R&D investments by leading pharmaceutical and biotechnology companies, focusing on novel drug development and personalized medicine, are contributing to market expansion. The expanding pipeline of treatments for specific LSDs like Gaucher disease, Pompe disease, Fabry disease, and cystinosis is also creating substantial opportunities for market players.
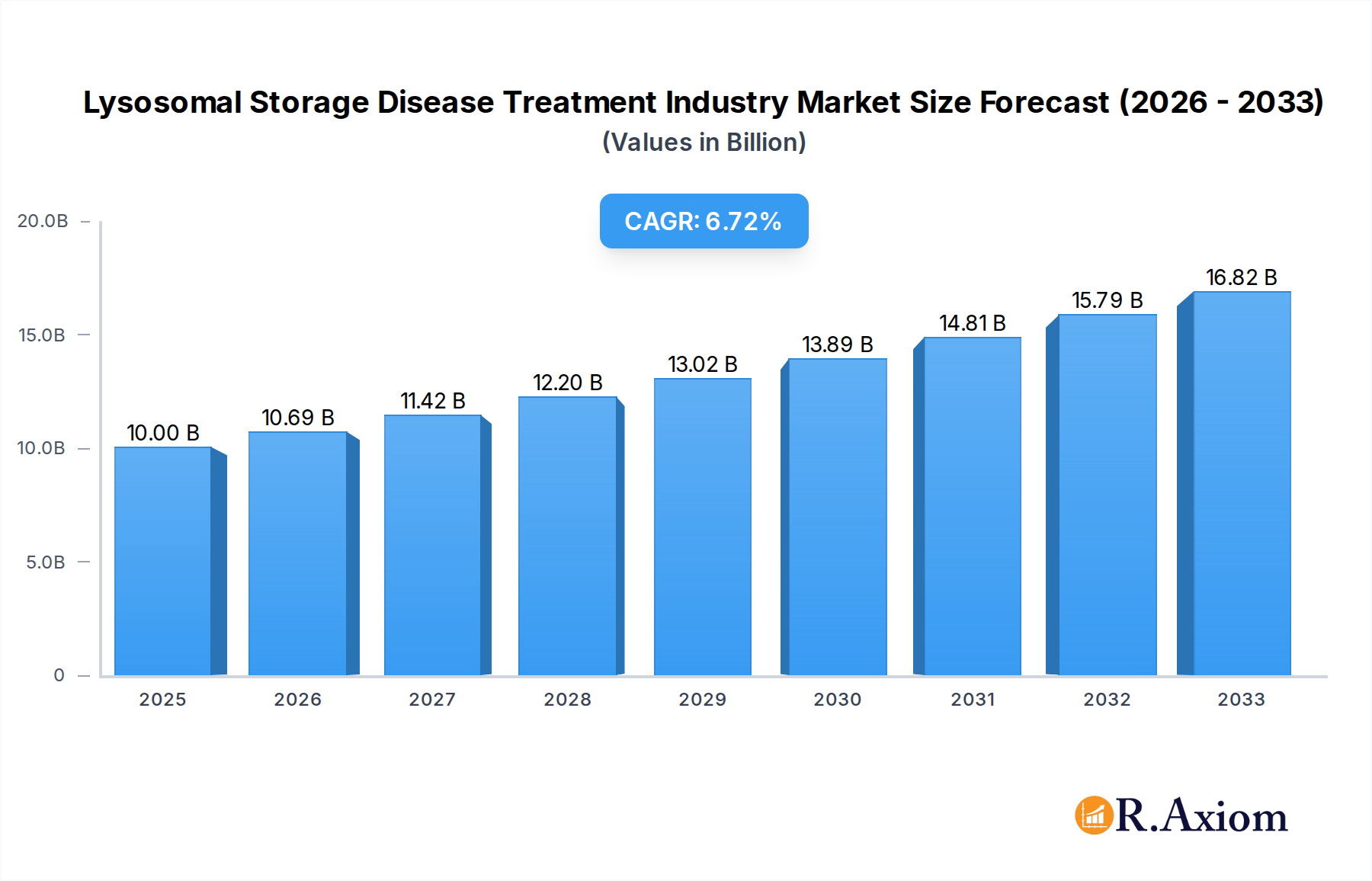
Lysosomal Storage Disease Treatment Industry Market Size (In Billion)

Despite the promising outlook, certain factors may temper the market's full potential. High treatment costs associated with specialized therapies, limited reimbursement policies in certain regions, and the complexity of drug development for rare diseases present significant challenges. Patient advocacy groups and increasing awareness initiatives are vital in driving demand and advocating for better access to these life-saving treatments. The market is also witnessing a trend towards the development of gene therapies and more targeted interventions, which, while promising long-term solutions, currently face regulatory hurdles and higher initial development costs. Collaborations between research institutions and pharmaceutical companies, along with strategic partnerships, are expected to accelerate innovation and broaden treatment accessibility, ultimately shaping the future landscape of LSD treatment.
Lysosomal Storage Disease Treatment Industry Company Market Share

Here is an SEO-optimized, detailed report description for the Lysosomal Storage Disease Treatment Industry, incorporating high-traffic keywords and adhering to all specified requirements:
Lysosomal Storage Disease Treatment Industry Market Concentration & Innovation
The Lysosomal Storage Disease (LSD) treatment market, projected to reach over $10 billion by 2033, exhibits a moderate level of concentration driven by a few dominant players. Innovation is the primary catalyst for growth, with significant investments in enzyme replacement therapy (ERT) and substrate reduction therapy (SRT). Regulatory frameworks, though evolving, present both opportunities and challenges for novel treatments, especially for rare genetic disorders. Product substitutes are limited due to the specific nature of LSDs, but advancements in gene therapy and small molecule drugs are emerging. End-user trends indicate a growing demand for personalized medicine and improved quality of life for patients. Mergers and acquisitions (M&A) are prevalent, with recent deals valued in the hundreds of millions of dollars, indicating a strategic consolidation of specialized expertise and product pipelines. For instance, the acquisition of Shire Plc by Takeda Pharmaceutical Company Limited for over $60 billion significantly reshaped the landscape. Key players like Sanofi (Genzyme Corporation), BioMarin, and Alexion Pharmaceuticals Inc. are consistently investing in R&D, holding substantial market share in specific LSD segments like Gaucher disease and Fabry disease.
Lysosomal Storage Disease Treatment Industry Industry Trends & Insights
The Lysosomal Storage Disease (LSD) treatment industry is poised for substantial growth, projected to achieve a compound annual growth rate (CAGR) exceeding 12% from 2025 to 2033. This robust expansion is fueled by an increasing incidence of rare genetic disorders, enhanced diagnostic capabilities, and significant advancements in therapeutic modalities. The market penetration of existing therapies is expanding, while novel treatment approaches are continuously entering the pipeline. Technological disruptions, particularly in gene therapy and advanced molecular targeting, are revolutionizing the treatment paradigm, offering potential cures rather than just symptom management for conditions like Pompe disease and Cystinosis. Consumer preferences are shifting towards precision medicine, demanding treatments tailored to individual genetic profiles and offering improved efficacy and safety profiles. Competitive dynamics are intense, with major pharmaceutical companies and emerging biotech firms vying for leadership. Companies are focusing on unmet medical needs within specific LSDs, leading to a diversification of the treatment landscape. The increasing prevalence of LSDs, estimated to affect approximately 1 in 5,000 live births, coupled with growing global healthcare expenditure, further propels market expansion. Furthermore, supportive government initiatives and orphan drug designations play a crucial role in incentivizing research and development for these rare conditions. The overall market is characterized by a strong emphasis on R&D investment and strategic partnerships to accelerate drug discovery and commercialization.
Dominant Markets & Segments in Lysosomal Storage Disease Treatment Industry
The global Lysosomal Storage Disease (LSD) treatment market demonstrates distinct regional dominance and segment leadership. North America, particularly the United States, currently holds the largest market share, driven by robust healthcare infrastructure, high patient awareness, and significant investments in rare disease research. Favorable reimbursement policies and the presence of leading pharmaceutical companies like BioMarin and Pfizer Inc. further solidify its position. Europe follows closely, with countries like Germany, the UK, and France showing strong market penetration due to advanced healthcare systems and government support for rare disease treatments.
Within the Therapy Type segmentation, Enzyme Replacement Therapy (ERT) currently dominates the market. This is primarily due to its established efficacy and widespread availability for conditions like Gaucher disease and Pompe disease. Companies such as Sanofi (Genzyme Corporation) and Amicus Therapeutics Inc. have established strong portfolios in ERT. However, Substrate Reduction Therapy (SRT) is emerging as a significant growth segment, offering a complementary or alternative approach for certain LSDs by reducing the production of accumulating substrates.
Analyzing Application segments, Gaucher disease remains a cornerstone of the LSD market, benefiting from well-established ERT treatments. Fabry disease is another major application, with ongoing advancements in both ERT and small molecule therapies. Pompe disease is witnessing considerable growth due to new therapeutic developments and increased diagnostic rates. Cystinosis, while rarer, is also seeing a steady rise in treatment demand as diagnostic tools improve. "Other Applications" encompass a broad range of less prevalent LSDs, presenting significant untapped potential for future therapeutic development. Key drivers for dominance in these segments include:
- Economic Policies: Government incentives for rare disease drug development, such as tax credits and extended market exclusivity, significantly impact market growth and accessibility.
- Infrastructure: Advanced diagnostic facilities, specialized treatment centers, and robust distribution networks are crucial for reaching patients with rare conditions.
- Regulatory Support: Streamlined approval pathways for orphan drugs by regulatory bodies like the FDA and EMA accelerate market entry for novel therapies.
- Patient Advocacy: Strong patient advocacy groups play a vital role in raising awareness, funding research, and influencing policy decisions.
Lysosomal Storage Disease Treatment Industry Product Developments
Product development in the Lysosomal Storage Disease (LSD) treatment industry is characterized by a strong focus on enhancing efficacy, improving patient convenience, and addressing unmet medical needs. Innovations include novel enzyme replacement therapies with extended half-lives, reducing the frequency of infusions. Gene therapy approaches are showing immense promise, offering the potential for long-term or even curative treatments for certain LSDs. Small molecule drugs targeting specific metabolic pathways are also gaining traction, providing oral administration options. These developments aim to offer greater convenience, reduced treatment burden, and improved patient outcomes, thereby creating significant competitive advantages for companies leading these advancements.
Report Scope & Segmentation Analysis
This report comprehensively analyzes the Lysosomal Storage Disease Treatment Industry, segmenting the market across key dimensions to provide detailed insights into market dynamics. The Therapy Type segmentation includes Enzyme Replacement Therapy (ERT) and Substrate Reduction Therapy (SRT), offering distinct treatment modalities for various LSDs. The Application segmentation covers prominent LSDs such as Gaucher disease, Cystinosis, Pompe Disease, and Fabry Disease, along with a category for Other Applications, encompassing a wide spectrum of rare genetic disorders. Each segment is analyzed for its current market size, projected growth, and competitive landscape, with ERT currently holding a larger market share but SRT exhibiting strong growth potential.
Key Drivers of Lysosomal Storage Disease Treatment Industry Growth
Several key factors are propelling the growth of the Lysosomal Storage Disease (LSD) Treatment Industry. Enhanced diagnostic capabilities, including newborn screening programs and advanced genetic testing, are identifying more patients earlier, thus increasing the demand for treatments. The increasing incidence of rare genetic disorders, coupled with growing global healthcare expenditure, provides a fertile ground for market expansion. Supportive regulatory frameworks, such as orphan drug designations and expedited review processes, incentivize pharmaceutical companies to invest in the research and development of treatments for these rare conditions. Furthermore, significant advancements in biotechnological research, particularly in gene therapy and targeted small molecule development, are leading to the introduction of more effective and innovative therapies, driving market growth.
Challenges in the Lysosomal Storage Disease Treatment Industry Sector
Despite the promising growth trajectory, the Lysosomal Storage Disease (LSD) Treatment Industry faces significant challenges. The high cost of developing and manufacturing treatments for rare diseases, often running into billions of dollars, translates into exorbitant drug prices, posing a major accessibility barrier for many patients. Stringent and lengthy regulatory approval processes for novel therapies, even for life-threatening conditions, can delay market entry. Furthermore, the limited patient population for each specific LSD makes it difficult for companies to achieve economies of scale in manufacturing and distribution, contributing to supply chain complexities. Intense competition among a few major players in established segments and the ongoing need for substantial R&D investment to address unmet needs also present ongoing challenges.
Emerging Opportunities in Lysosomal Storage Disease Treatment Industry
The Lysosomal Storage Disease (LSD) Treatment Industry is ripe with emerging opportunities. The development and adoption of gene therapy represent a significant frontier, offering the potential for one-time, curative treatments for many LSDs, opening up vast market potential. Advancements in precision medicine and personalized therapeutic approaches, driven by sophisticated genetic sequencing and data analytics, allow for the tailoring of treatments to individual patient profiles, enhancing efficacy. The increasing global focus on rare diseases, supported by government initiatives and patient advocacy groups, is creating new market opportunities and fostering collaborative research. Furthermore, the untapped potential in developing treatments for less common LSDs presents a significant opportunity for innovation and market expansion, with many new drugs in the pipeline for these conditions.
Leading Players in the Lysosomal Storage Disease Treatment Industry Market
- Sanofi
- Amicus Therapeutics Inc
- Sigilon Therapeutics Inc
- BioMarin
- Alexion Pharmaceuticals Inc
- Takeda Pharmaceutical Company Limited
- Orphazyme A/S
- Johnson & Johnson
- Pfizer Inc
Key Developments in Lysosomal Storage Disease Treatment Industry Industry
- August 2022: The European Commission approved Galafold, marketed by Amicus Therapeutics, for the long-term treatment of Fabry disease patients aged 12 years and above with an amenable mutation. This approval expands treatment options for Fabry disease in a key European market.
- February 2022: Maze Therapeutics reported new preclinical data supporting the advancement of MZE001. This investigational therapy aims to address Pompe disease by reducing pathologic glycogen accumulation through the inhibition of muscle glycogen synthase (GYS1), offering a novel therapeutic mechanism for this debilitating condition.
Strategic Outlook for Lysosomal Storage Disease Treatment Industry Market
The strategic outlook for the Lysosomal Storage Disease (LSD) Treatment Industry remains exceptionally positive, driven by persistent innovation and a growing understanding of rare genetic disorders. The market is expected to witness sustained growth, fueled by advancements in gene therapy, small molecule development, and improved diagnostic tools. Emphasis on personalized medicine and patient-centric care will continue to shape product development and market strategies. Strategic collaborations and partnerships between pharmaceutical giants and nimble biotech firms will be crucial for accelerating R&D and expanding market reach. The increasing global awareness and supportive regulatory environments for orphan drugs further enhance the long-term growth prospects, promising significant returns for stakeholders investing in this critical therapeutic area.
Lysosomal Storage Disease Treatment Industry Segmentation
-
1. Therapy Type
- 1.1. Enzyme Replacement Therapy
- 1.2. Substrate Reduction Therapy
-
2. Application
- 2.1. Gaucher disease
- 2.2. Cystinosis
- 2.3. Pompe Disease
- 2.4. Fabry Disease
- 2.5. Other Applications
Lysosomal Storage Disease Treatment Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
- 4. Rest of the World
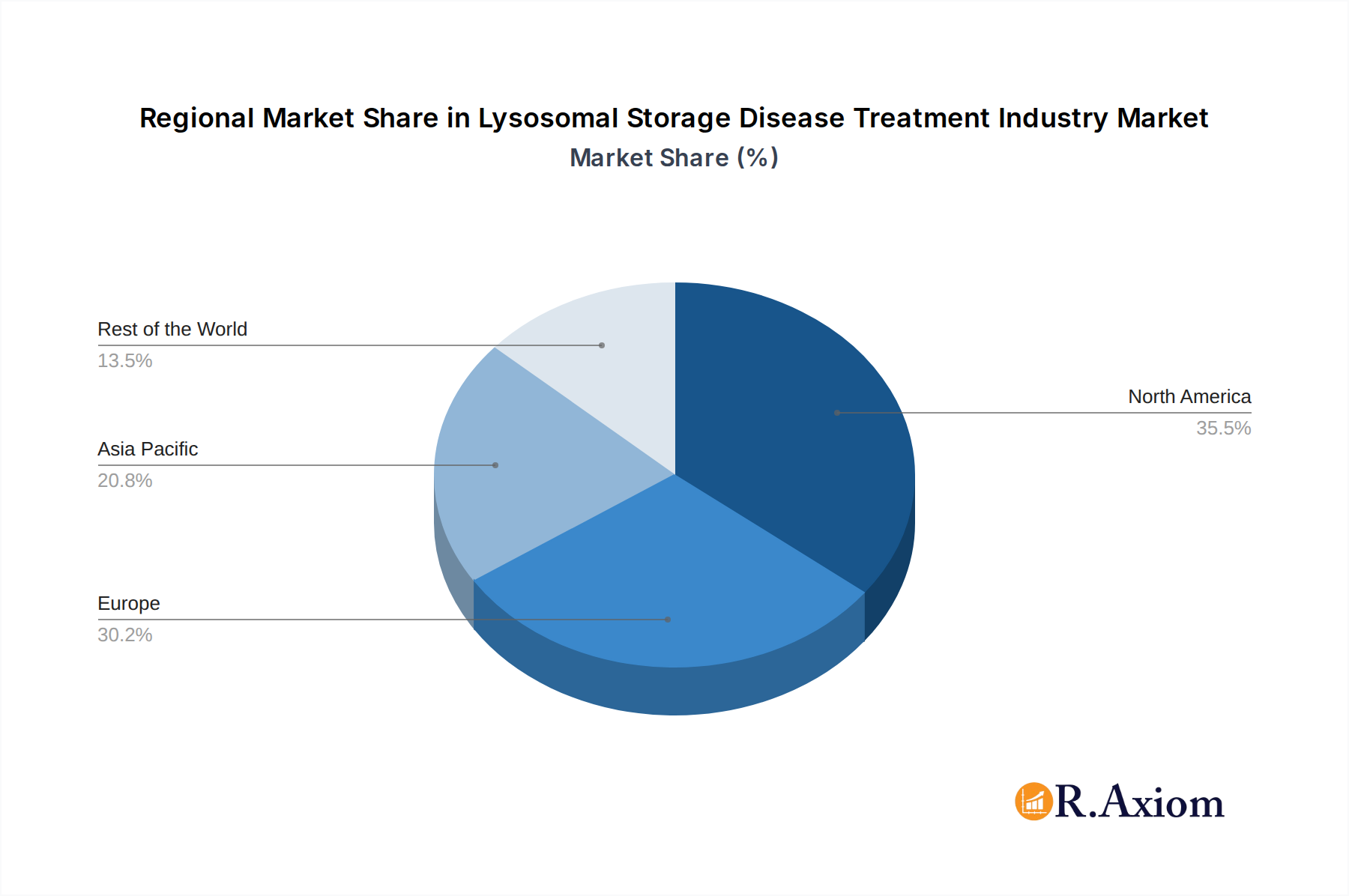
Lysosomal Storage Disease Treatment Industry Regional Market Share

Geographic Coverage of Lysosomal Storage Disease Treatment Industry
Lysosomal Storage Disease Treatment Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.9% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. RAX Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Therapy Type
- 5.1.1. Enzyme Replacement Therapy
- 5.1.2. Substrate Reduction Therapy
- 5.2. Market Analysis, Insights and Forecast - by Application
- 5.2.1. Gaucher disease
- 5.2.2. Cystinosis
- 5.2.3. Pompe Disease
- 5.2.4. Fabry Disease
- 5.2.5. Other Applications
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. Europe
- 5.3.3. Asia Pacific
- 5.3.4. Rest of the World
- 5.1. Market Analysis, Insights and Forecast - by Therapy Type
- 6. Global Lysosomal Storage Disease Treatment Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Therapy Type
- 6.1.1. Enzyme Replacement Therapy
- 6.1.2. Substrate Reduction Therapy
- 6.2. Market Analysis, Insights and Forecast - by Application
- 6.2.1. Gaucher disease
- 6.2.2. Cystinosis
- 6.2.3. Pompe Disease
- 6.2.4. Fabry Disease
- 6.2.5. Other Applications
- 6.1. Market Analysis, Insights and Forecast - by Therapy Type
- 7. North America Lysosomal Storage Disease Treatment Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Therapy Type
- 7.1.1. Enzyme Replacement Therapy
- 7.1.2. Substrate Reduction Therapy
- 7.2. Market Analysis, Insights and Forecast - by Application
- 7.2.1. Gaucher disease
- 7.2.2. Cystinosis
- 7.2.3. Pompe Disease
- 7.2.4. Fabry Disease
- 7.2.5. Other Applications
- 7.1. Market Analysis, Insights and Forecast - by Therapy Type
- 8. Europe Lysosomal Storage Disease Treatment Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Therapy Type
- 8.1.1. Enzyme Replacement Therapy
- 8.1.2. Substrate Reduction Therapy
- 8.2. Market Analysis, Insights and Forecast - by Application
- 8.2.1. Gaucher disease
- 8.2.2. Cystinosis
- 8.2.3. Pompe Disease
- 8.2.4. Fabry Disease
- 8.2.5. Other Applications
- 8.1. Market Analysis, Insights and Forecast - by Therapy Type
- 9. Asia Pacific Lysosomal Storage Disease Treatment Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Therapy Type
- 9.1.1. Enzyme Replacement Therapy
- 9.1.2. Substrate Reduction Therapy
- 9.2. Market Analysis, Insights and Forecast - by Application
- 9.2.1. Gaucher disease
- 9.2.2. Cystinosis
- 9.2.3. Pompe Disease
- 9.2.4. Fabry Disease
- 9.2.5. Other Applications
- 9.1. Market Analysis, Insights and Forecast - by Therapy Type
- 10. Rest of the World Lysosomal Storage Disease Treatment Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Therapy Type
- 10.1.1. Enzyme Replacement Therapy
- 10.1.2. Substrate Reduction Therapy
- 10.2. Market Analysis, Insights and Forecast - by Application
- 10.2.1. Gaucher disease
- 10.2.2. Cystinosis
- 10.2.3. Pompe Disease
- 10.2.4. Fabry Disease
- 10.2.5. Other Applications
- 10.1. Market Analysis, Insights and Forecast - by Therapy Type
- 11. Competitive Analysis
- 11.1. Company Profiles
- 11.1.1 Sanofi (Genzyme Corporation)
- 11.1.1.1. Company Overview
- 11.1.1.2. Products
- 11.1.1.3. Company Financials
- 11.1.1.4. SWOT Analysis
- 11.1.2 Amicus Therapeutics Inc
- 11.1.2.1. Company Overview
- 11.1.2.2. Products
- 11.1.2.3. Company Financials
- 11.1.2.4. SWOT Analysis
- 11.1.3 Sigilon Therapeutics Inc
- 11.1.3.1. Company Overview
- 11.1.3.2. Products
- 11.1.3.3. Company Financials
- 11.1.3.4. SWOT Analysis
- 11.1.4 BioMarin
- 11.1.4.1. Company Overview
- 11.1.4.2. Products
- 11.1.4.3. Company Financials
- 11.1.4.4. SWOT Analysis
- 11.1.5 Alexion Pharmaceuticals Inc
- 11.1.5.1. Company Overview
- 11.1.5.2. Products
- 11.1.5.3. Company Financials
- 11.1.5.4. SWOT Analysis
- 11.1.6 Takeda Pharmaceutical Company Limited (Shire Plc)
- 11.1.6.1. Company Overview
- 11.1.6.2. Products
- 11.1.6.3. Company Financials
- 11.1.6.4. SWOT Analysis
- 11.1.7 Orphazyme A/S
- 11.1.7.1. Company Overview
- 11.1.7.2. Products
- 11.1.7.3. Company Financials
- 11.1.7.4. SWOT Analysis
- 11.1.8 Johnson & Johnson (Actelion Pharmaceuticals Ltd)
- 11.1.8.1. Company Overview
- 11.1.8.2. Products
- 11.1.8.3. Company Financials
- 11.1.8.4. SWOT Analysis
- 11.1.9 Pfizer Inc
- 11.1.9.1. Company Overview
- 11.1.9.2. Products
- 11.1.9.3. Company Financials
- 11.1.9.4. SWOT Analysis
- 11.1.1 Sanofi (Genzyme Corporation)
- 11.2. Market Entropy
- 11.2.1 Company's Key Areas Served
- 11.2.2 Recent Developments
- 11.3. Company Market Share Analysis 2025
- 11.3.1 Top 5 Companies Market Share Analysis
- 11.3.2 Top 3 Companies Market Share Analysis
- 11.4. List of Potential Customers
- 12. Research Methodology
List of Figures
- Figure 1: Global Lysosomal Storage Disease Treatment Industry Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: Global Lysosomal Storage Disease Treatment Industry Volume Breakdown (K Unit, %) by Region 2025 & 2033
- Figure 3: North America Lysosomal Storage Disease Treatment Industry Revenue (billion), by Therapy Type 2025 & 2033
- Figure 4: North America Lysosomal Storage Disease Treatment Industry Volume (K Unit), by Therapy Type 2025 & 2033
- Figure 5: North America Lysosomal Storage Disease Treatment Industry Revenue Share (%), by Therapy Type 2025 & 2033
- Figure 6: North America Lysosomal Storage Disease Treatment Industry Volume Share (%), by Therapy Type 2025 & 2033
- Figure 7: North America Lysosomal Storage Disease Treatment Industry Revenue (billion), by Application 2025 & 2033
- Figure 8: North America Lysosomal Storage Disease Treatment Industry Volume (K Unit), by Application 2025 & 2033
- Figure 9: North America Lysosomal Storage Disease Treatment Industry Revenue Share (%), by Application 2025 & 2033
- Figure 10: North America Lysosomal Storage Disease Treatment Industry Volume Share (%), by Application 2025 & 2033
- Figure 11: North America Lysosomal Storage Disease Treatment Industry Revenue (billion), by Country 2025 & 2033
- Figure 12: North America Lysosomal Storage Disease Treatment Industry Volume (K Unit), by Country 2025 & 2033
- Figure 13: North America Lysosomal Storage Disease Treatment Industry Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America Lysosomal Storage Disease Treatment Industry Volume Share (%), by Country 2025 & 2033
- Figure 15: Europe Lysosomal Storage Disease Treatment Industry Revenue (billion), by Therapy Type 2025 & 2033
- Figure 16: Europe Lysosomal Storage Disease Treatment Industry Volume (K Unit), by Therapy Type 2025 & 2033
- Figure 17: Europe Lysosomal Storage Disease Treatment Industry Revenue Share (%), by Therapy Type 2025 & 2033
- Figure 18: Europe Lysosomal Storage Disease Treatment Industry Volume Share (%), by Therapy Type 2025 & 2033
- Figure 19: Europe Lysosomal Storage Disease Treatment Industry Revenue (billion), by Application 2025 & 2033
- Figure 20: Europe Lysosomal Storage Disease Treatment Industry Volume (K Unit), by Application 2025 & 2033
- Figure 21: Europe Lysosomal Storage Disease Treatment Industry Revenue Share (%), by Application 2025 & 2033
- Figure 22: Europe Lysosomal Storage Disease Treatment Industry Volume Share (%), by Application 2025 & 2033
- Figure 23: Europe Lysosomal Storage Disease Treatment Industry Revenue (billion), by Country 2025 & 2033
- Figure 24: Europe Lysosomal Storage Disease Treatment Industry Volume (K Unit), by Country 2025 & 2033
- Figure 25: Europe Lysosomal Storage Disease Treatment Industry Revenue Share (%), by Country 2025 & 2033
- Figure 26: Europe Lysosomal Storage Disease Treatment Industry Volume Share (%), by Country 2025 & 2033
- Figure 27: Asia Pacific Lysosomal Storage Disease Treatment Industry Revenue (billion), by Therapy Type 2025 & 2033
- Figure 28: Asia Pacific Lysosomal Storage Disease Treatment Industry Volume (K Unit), by Therapy Type 2025 & 2033
- Figure 29: Asia Pacific Lysosomal Storage Disease Treatment Industry Revenue Share (%), by Therapy Type 2025 & 2033
- Figure 30: Asia Pacific Lysosomal Storage Disease Treatment Industry Volume Share (%), by Therapy Type 2025 & 2033
- Figure 31: Asia Pacific Lysosomal Storage Disease Treatment Industry Revenue (billion), by Application 2025 & 2033
- Figure 32: Asia Pacific Lysosomal Storage Disease Treatment Industry Volume (K Unit), by Application 2025 & 2033
- Figure 33: Asia Pacific Lysosomal Storage Disease Treatment Industry Revenue Share (%), by Application 2025 & 2033
- Figure 34: Asia Pacific Lysosomal Storage Disease Treatment Industry Volume Share (%), by Application 2025 & 2033
- Figure 35: Asia Pacific Lysosomal Storage Disease Treatment Industry Revenue (billion), by Country 2025 & 2033
- Figure 36: Asia Pacific Lysosomal Storage Disease Treatment Industry Volume (K Unit), by Country 2025 & 2033
- Figure 37: Asia Pacific Lysosomal Storage Disease Treatment Industry Revenue Share (%), by Country 2025 & 2033
- Figure 38: Asia Pacific Lysosomal Storage Disease Treatment Industry Volume Share (%), by Country 2025 & 2033
- Figure 39: Rest of the World Lysosomal Storage Disease Treatment Industry Revenue (billion), by Therapy Type 2025 & 2033
- Figure 40: Rest of the World Lysosomal Storage Disease Treatment Industry Volume (K Unit), by Therapy Type 2025 & 2033
- Figure 41: Rest of the World Lysosomal Storage Disease Treatment Industry Revenue Share (%), by Therapy Type 2025 & 2033
- Figure 42: Rest of the World Lysosomal Storage Disease Treatment Industry Volume Share (%), by Therapy Type 2025 & 2033
- Figure 43: Rest of the World Lysosomal Storage Disease Treatment Industry Revenue (billion), by Application 2025 & 2033
- Figure 44: Rest of the World Lysosomal Storage Disease Treatment Industry Volume (K Unit), by Application 2025 & 2033
- Figure 45: Rest of the World Lysosomal Storage Disease Treatment Industry Revenue Share (%), by Application 2025 & 2033
- Figure 46: Rest of the World Lysosomal Storage Disease Treatment Industry Volume Share (%), by Application 2025 & 2033
- Figure 47: Rest of the World Lysosomal Storage Disease Treatment Industry Revenue (billion), by Country 2025 & 2033
- Figure 48: Rest of the World Lysosomal Storage Disease Treatment Industry Volume (K Unit), by Country 2025 & 2033
- Figure 49: Rest of the World Lysosomal Storage Disease Treatment Industry Revenue Share (%), by Country 2025 & 2033
- Figure 50: Rest of the World Lysosomal Storage Disease Treatment Industry Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Therapy Type 2020 & 2033
- Table 2: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Therapy Type 2020 & 2033
- Table 3: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 4: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Application 2020 & 2033
- Table 5: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Region 2020 & 2033
- Table 6: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Region 2020 & 2033
- Table 7: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Therapy Type 2020 & 2033
- Table 8: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Therapy Type 2020 & 2033
- Table 9: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 10: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Application 2020 & 2033
- Table 11: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 12: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 13: United States Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: United States Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 15: Canada Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Canada Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 17: Mexico Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 18: Mexico Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 19: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Therapy Type 2020 & 2033
- Table 20: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Therapy Type 2020 & 2033
- Table 21: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 22: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Application 2020 & 2033
- Table 23: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 24: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 25: Germany Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: Germany Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 27: United Kingdom Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: United Kingdom Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 29: France Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 30: France Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 31: Italy Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: Italy Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 33: Spain Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 34: Spain Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 35: Rest of Europe Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 36: Rest of Europe Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 37: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Therapy Type 2020 & 2033
- Table 38: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Therapy Type 2020 & 2033
- Table 39: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 40: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Application 2020 & 2033
- Table 41: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 42: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 43: China Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: China Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 45: Japan Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 46: Japan Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 47: India Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 48: India Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 49: Australia Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 50: Australia Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 51: South Korea Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 52: South Korea Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 53: Rest of Asia Pacific Lysosomal Storage Disease Treatment Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 54: Rest of Asia Pacific Lysosomal Storage Disease Treatment Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 55: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Therapy Type 2020 & 2033
- Table 56: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Therapy Type 2020 & 2033
- Table 57: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 58: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Application 2020 & 2033
- Table 59: Global Lysosomal Storage Disease Treatment Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 60: Global Lysosomal Storage Disease Treatment Industry Volume K Unit Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Lysosomal Storage Disease Treatment Industry?
The projected CAGR is approximately 6.9%.
2. Which companies are prominent players in the Lysosomal Storage Disease Treatment Industry?
Key companies in the market include Sanofi (Genzyme Corporation), Amicus Therapeutics Inc, Sigilon Therapeutics Inc, BioMarin, Alexion Pharmaceuticals Inc, Takeda Pharmaceutical Company Limited (Shire Plc), Orphazyme A/S, Johnson & Johnson (Actelion Pharmaceuticals Ltd), Pfizer Inc.
3. What are the main segments of the Lysosomal Storage Disease Treatment Industry?
The market segments include Therapy Type, Application.
4. Can you provide details about the market size?
The market size is estimated to be USD 10 billion as of 2022.
5. What are some drivers contributing to market growth?
Increasing Incidences of Lysosomal Diseases and Growing Awrness; Increasing Research and Development for Diagnosis and Drug Development for Treatment of Lysosomal Diseases.
6. What are the notable trends driving market growth?
Pompe Disease Segment is Expected to Hold a Major Market Share in the Lysosomal Storage Disease Treatment Market.
7. Are there any restraints impacting market growth?
High Cost of Treatment.
8. Can you provide examples of recent developments in the market?
In August 2022, the European Commission approved Galafold marketed by Amicus Therapeutics for the long-term treatment of Fabry disease patients aged 12 years and above with an amenable mutation.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Lysosomal Storage Disease Treatment Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Lysosomal Storage Disease Treatment Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Lysosomal Storage Disease Treatment Industry?
To stay informed about further developments, trends, and reports in the Lysosomal Storage Disease Treatment Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
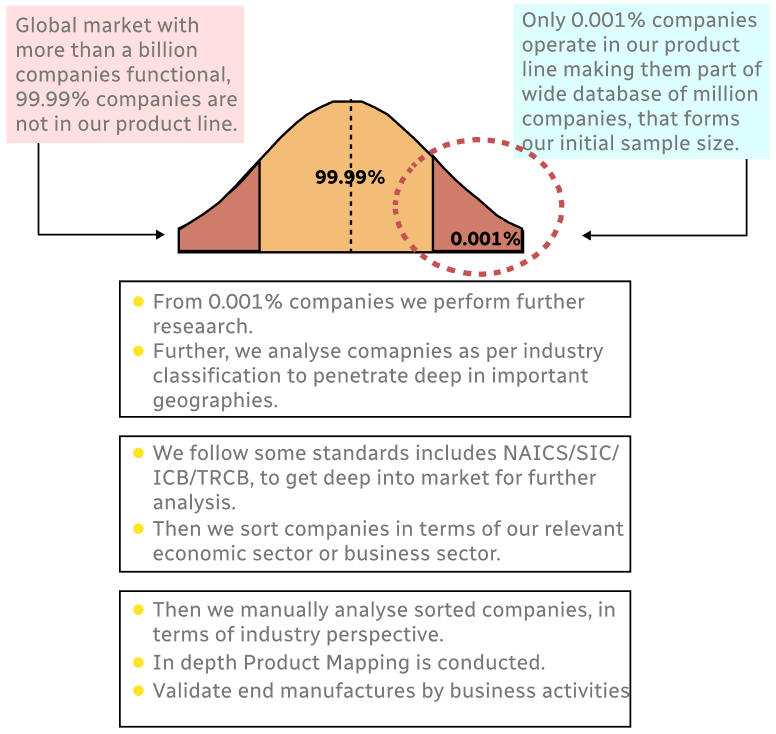
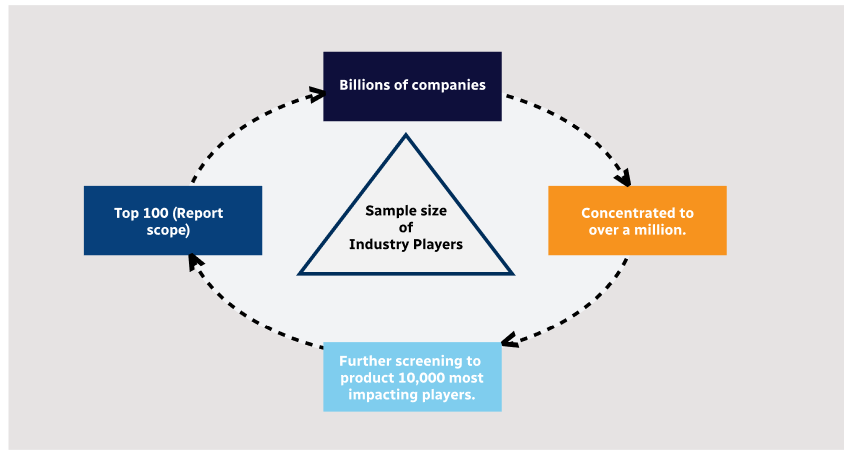
Step 1 - Identification of Relevant Samples Size from Population Database
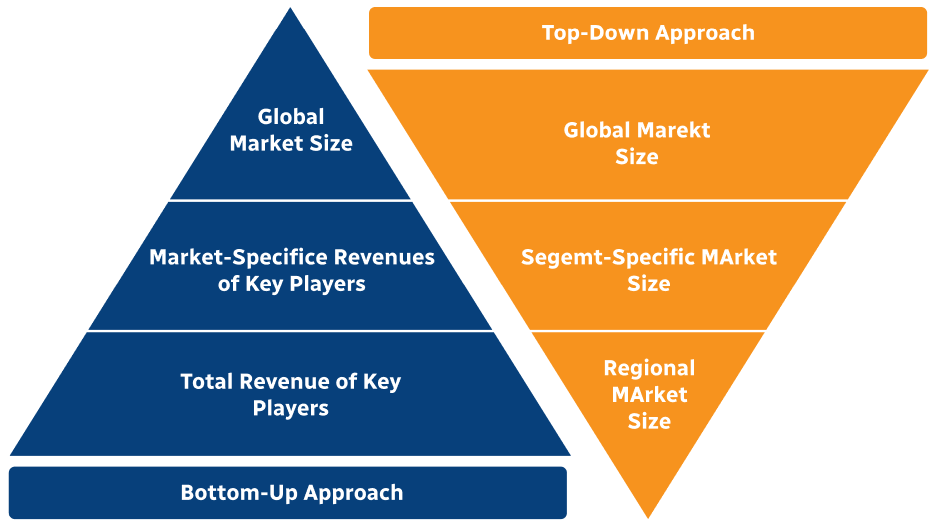
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence