Key Insights
The North America Cardiac Rhythm Monitoring Industry is poised for substantial growth, projected to reach $30.05 billion in 2025. This dynamic market is driven by an increasing prevalence of cardiovascular diseases, a growing aging population, and significant advancements in cardiac monitoring technologies. The estimated Compound Annual Growth Rate (CAGR) of 6.55% for the forecast period (2025-2033) underscores the industry's robust expansion. Key market drivers include rising awareness of cardiac health, greater adoption of wearable and implantable devices for continuous monitoring, and favorable reimbursement policies for advanced cardiac care. The industry is segmented into crucial product categories: Defibrillators (including Implantable Cardioverter Defibrillators - ICDs and External Defibrillators - EDs), Pacemakers (Implantable Pacemakers - ICPs and External Pacemakers), and Cardiac Resynchronization Therapy (CRT) Devices. These segments are experiencing innovation, with a focus on miniaturization, enhanced efficacy, and improved patient compliance.
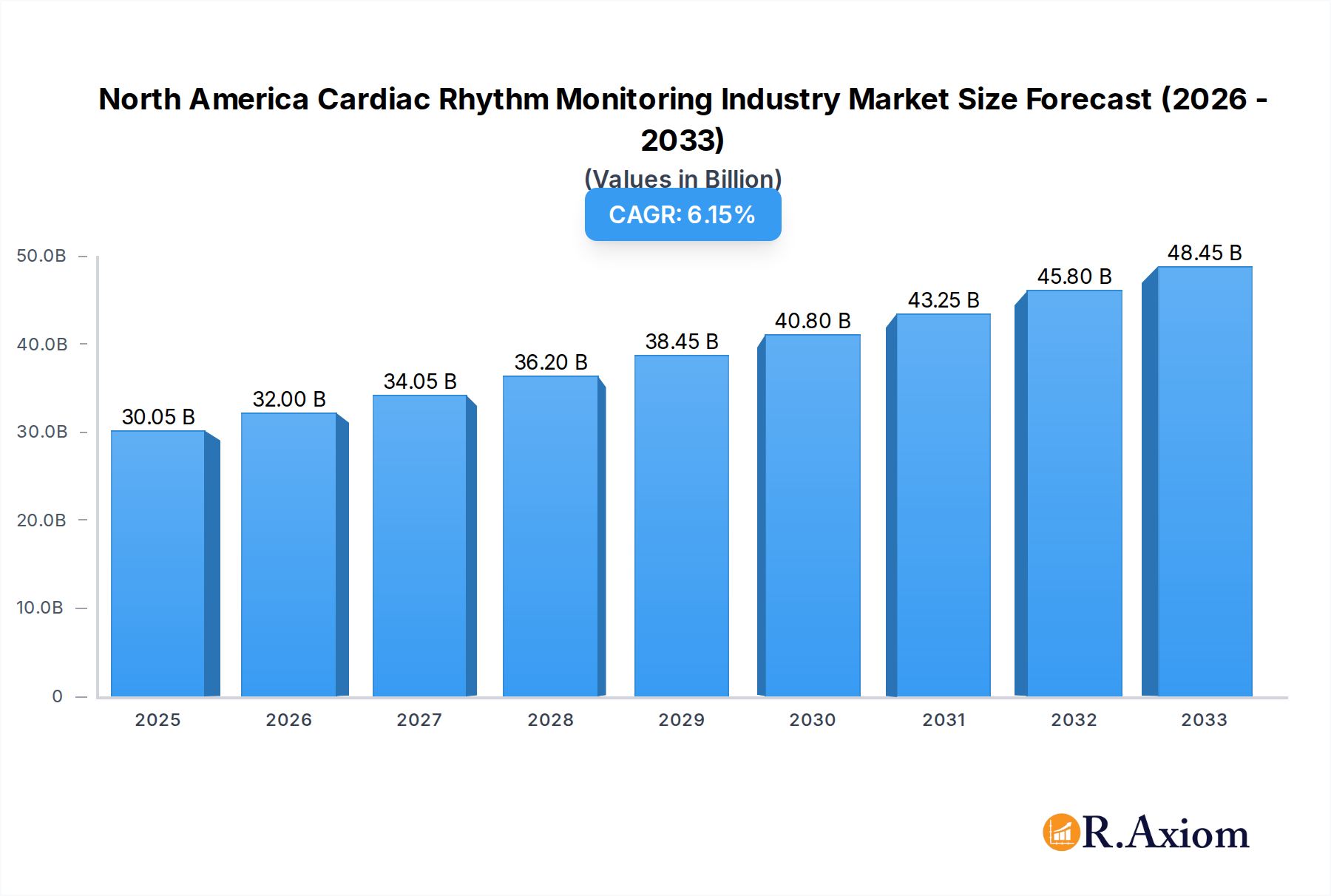
North America Cardiac Rhythm Monitoring Industry Market Size (In Billion)

The market landscape is characterized by significant investment in research and development by leading global companies such as Medtronic PLC, Abbott Laboratories, Boston Scientific Corporation, and Koninklijke Philips NV. These players are at the forefront of introducing novel solutions for diagnosing and treating cardiac arrhythmias. Emerging trends include the integration of artificial intelligence (AI) and machine learning (ML) for predictive analytics in cardiac monitoring, the expansion of remote patient monitoring services, and the development of leadless pacemakers and smaller ICDs. While the market exhibits strong growth potential, certain restraints, such as high device costs and regulatory hurdles, need to be addressed. Geographically, the United States dominates the North American market due to its advanced healthcare infrastructure, high patient spending, and strong R&D ecosystem. Canada and Mexico are also expected to witness steady growth, driven by increasing healthcare expenditure and a rising incidence of cardiac conditions.
North America Cardiac Rhythm Monitoring Industry Company Market Share

North America Cardiac Rhythm Monitoring Industry Market Concentration & Innovation
The North America Cardiac Rhythm Monitoring Industry is characterized by a moderate to high market concentration, with a few key players dominating the landscape. Major companies like Medtronic PLC, Abbott Laboratories, Boston Scientific Corporation, and Koninklijke Philips NV hold significant market share, driven by their extensive product portfolios and strong research and development capabilities. Innovation is a critical differentiator, with continuous advancements in miniaturization, battery life, data analytics, and remote monitoring technologies fueling market growth. Regulatory frameworks, particularly stringent FDA approvals in the United States, act as both a barrier to entry and a driver for innovative and compliant solutions. Product substitutes are limited in the core cardiac rhythm monitoring space, but advancements in wearable technology and AI-driven diagnostics present emerging indirect competition. End-user trends favor less invasive and more patient-centric monitoring solutions, pushing the demand for implantable devices and advanced wearable monitors. Mergers and acquisitions (M&A) activity, while not always headline-grabbing, are strategic moves for companies seeking to expand their technological capabilities, market reach, or acquire niche expertise. M&A deal values in this specialized sector can range from tens of millions to billions of dollars for significant acquisitions, impacting market concentration and innovation pathways.
North America Cardiac Rhythm Monitoring Industry Industry Trends & Insights
The North America Cardiac Rhythm Monitoring Industry is experiencing robust growth, projected to witness a Compound Annual Growth Rate (CAGR) of approximately 7.5% from 2025 to 2033. This expansion is primarily driven by the increasing prevalence of cardiovascular diseases, including atrial fibrillation, heart failure, and sudden cardiac arrest, which necessitate continuous and accurate rhythm monitoring. An aging global population, particularly in North America, further fuels demand as the risk of cardiac arrhythmias escalates with age. Technological advancements are revolutionizing the industry. The integration of artificial intelligence (AI) and machine learning (ML) into cardiac rhythm monitoring devices is enabling more sophisticated data analysis, predictive diagnostics, and early detection of critical events. This leads to improved patient outcomes and reduced healthcare costs. Remote patient monitoring (RPM) technologies are also gaining significant traction. These systems allow healthcare providers to continuously track patient data from a distance, enabling timely interventions and reducing the need for frequent hospital visits. This trend is further accelerated by the increasing adoption of telehealth services, especially post-pandemic. Consumer preferences are shifting towards user-friendly, non-invasive, and highly accurate devices. Patients are becoming more proactive in managing their health, leading to a demand for advanced wearable cardiac monitors and smart devices that can seamlessly integrate with their daily lives. The competitive landscape is intense, with established players like Medtronic PLC, Abbott Laboratories, and Boston Scientific Corporation investing heavily in R&D to maintain their market leadership. Emerging companies are focusing on niche areas, such as novel sensor technologies or specialized diagnostic algorithms. Market penetration of implantable cardiac devices is already high, but the growth in this segment is driven by next-generation devices with enhanced functionality and longevity. The external cardiac monitoring segment, including Holter monitors and event recorders, is also seeing growth due to increased awareness and technological improvements making these devices more accessible and informative. Furthermore, the increasing emphasis on preventative healthcare and early diagnosis by both healthcare providers and payers is a significant market driver. Initiatives aimed at reducing hospital readmissions for cardiac-related issues are also boosting the adoption of effective cardiac rhythm monitoring solutions. The ongoing evolution of diagnostic capabilities, moving from simple rhythm detection to sophisticated analysis of complex arrhythmias, is creating new market opportunities.
Dominant Markets & Segments in North America Cardiac Rhythm Monitoring Industry
The United States stands as the undisputed dominant market within the North America Cardiac Rhythm Monitoring Industry, commanding a substantial majority of the market share. This dominance is attributable to several intertwined factors, including a highly developed healthcare infrastructure, a large and aging population with a high incidence of cardiovascular diseases, significant investment in medical research and development, and a favorable reimbursement landscape for advanced medical technologies. Economic policies in the US actively encourage innovation and adoption of cutting-edge medical devices, further bolstering the market. Robust regulatory frameworks, while stringent, also create a structured environment for product approval and market entry for credible manufacturers.
Within the United States, the Implantable Cardioverter Defibrillators (ICDs) segment exhibits significant dominance. The high prevalence of life-threatening arrhythmias, coupled with a proactive approach to managing patients at risk for sudden cardiac arrest, drives the demand for ICDs. Advanced features such as S-ICD (subcutaneous ICD) systems, offering less invasive implantation, and ICDs with advanced diagnostic capabilities are further fueling growth in this segment. Implantable Pacemakers (IPCs) also represent a substantial and growing segment, driven by the increasing incidence of bradycardia and heart block, particularly in an aging demographic. The development of smaller, longer-lasting pacemakers with sophisticated pacing algorithms contributes to their market penetration.
The External Defibrillators (ED) market, particularly automated external defibrillators (AEDs), is witnessing steady growth, propelled by public access defibrillation programs and increased awareness of their life-saving potential in out-of-hospital settings. Government initiatives and corporate social responsibility programs are key drivers for AED deployment in public spaces. Similarly, the External Pacemakers segment, though smaller than its implantable counterpart, serves critical functions in temporary pacing during acute cardiac events or post-surgery.
Cardiac Resynchronization Therapy (CRT) devices, encompassing both CRT-Defibrillators (CRT-D) and CRT-Pacemakers (CRT-P), are crucial for managing heart failure patients with conduction abnormalities. The growing burden of heart failure in North America, especially in the United States, makes this a high-impact and growing segment. Technological advancements leading to improved CRT programming and patient selection are enhancing its effectiveness and market appeal. The development of CRT devices that offer a combined therapy for both heart failure and arrhythmias is a key innovation driving segment growth.
Canada and Mexico, while smaller markets compared to the United States, are also experiencing growth in cardiac rhythm monitoring. Canada benefits from a universal healthcare system that supports the adoption of advanced medical technologies, while Mexico's market is expanding due to an increasing focus on improving healthcare access and the growing prevalence of cardiovascular diseases. Economic development and increasing private healthcare investment in Mexico are key drivers for its expanding market.
North America Cardiac Rhythm Monitoring Industry Product Developments
Product innovation in the North America Cardiac Rhythm Monitoring Industry centers on enhancing diagnostic accuracy, improving patient comfort, and enabling seamless remote monitoring. Manufacturers are focusing on developing smaller, less invasive implantable devices with extended battery life, such as insertable cardiac monitors (ICMs) that can continuously track arrhythmias for up to three years. Advancements in sensing technology are leading to more precise detection of subtle rhythm abnormalities, crucial for diagnosing conditions like atrial fibrillation and cryptogenic stroke. Furthermore, the integration of AI and machine learning algorithms into these devices allows for intelligent data analysis, providing actionable insights to clinicians and enabling proactive patient management. The development of wireless data transmission capabilities and cloud-based platforms is crucial for facilitating remote patient monitoring, thereby improving patient outcomes and reducing healthcare burdens.
Report Scope & Segmentation Analysis
This report provides a comprehensive analysis of the North America Cardiac Rhythm Monitoring Industry, segmented by product type and geography. Product segmentation includes Defibrillators, further categorized into Implantable Cardioverter Defibrillators (ICDS) and External Defibrillators (ED). Pacemakers are segmented into Implantable Pacemakers (ICPS) and External Pacemakers. Cardiac Resynchronization Therapy Devices are analyzed as a distinct category. Geographically, the market is divided into the United States, Canada, and Mexico. The United States is expected to continue its dominance, driven by a large patient pool and advanced healthcare infrastructure, with strong growth projected for ICDs and CRT devices. Canada's market is anticipated to see steady expansion, supported by its healthcare system. Mexico represents a high-growth opportunity, with increasing healthcare investments and a rising incidence of cardiac conditions. The forecast period from 2025 to 2033 will witness significant technological evolution across all segments.
Key Drivers of North America Cardiac Rhythm Monitoring Industry Growth
The North America Cardiac Rhythm Monitoring Industry is propelled by several key drivers. The escalating prevalence of cardiovascular diseases, particularly atrial fibrillation and heart failure, due to an aging population and lifestyle factors, creates a sustained demand for effective monitoring solutions. Technological advancements, including miniaturization, enhanced data analytics capabilities powered by AI, and sophisticated remote monitoring platforms, are critical in improving diagnostic accuracy and patient management. Furthermore, favorable reimbursement policies and government initiatives aimed at improving patient outcomes and reducing healthcare costs encourage the adoption of these advanced technologies. Increased patient awareness and a growing demand for proactive health management also contribute significantly to market expansion.
Challenges in the North America Cardiac Rhythm Monitoring Industry Sector
Despite its robust growth, the North America Cardiac Rhythm Monitoring Industry faces several challenges. Stringent regulatory approval processes, particularly from the U.S. Food and Drug Administration (FDA), can prolong product development timelines and increase costs. The high cost of advanced cardiac rhythm monitoring devices can be a barrier to widespread adoption, especially in healthcare systems with budget constraints. Cybersecurity concerns related to the transmission and storage of sensitive patient data also pose a significant challenge, requiring robust security measures. Intense competition among established players and emerging innovators can lead to price pressures and a constant need for differentiation. Moreover, the need for specialized training for healthcare professionals to effectively utilize and interpret data from complex monitoring devices can also present a hurdle.
Emerging Opportunities in North America Cardiac Rhythm Monitoring Industry
Emerging opportunities in the North America Cardiac Rhythm Monitoring Industry are abundant, driven by technological innovation and evolving healthcare paradigms. The widespread adoption of telehealth and remote patient monitoring (RPM) presents a significant avenue for growth, allowing for continuous patient oversight and early intervention. The integration of Artificial Intelligence (AI) and Machine Learning (ML) in diagnostic algorithms offers the potential for predictive analytics, enabling the detection of arrhythmias before they become critical. Advances in wearable technology are creating opportunities for non-invasive and user-friendly monitoring solutions that can be seamlessly integrated into daily life. The development of novel therapeutic devices that combine monitoring and treatment capabilities also represents a promising area for innovation and market expansion. Furthermore, exploring underdeveloped markets within North America and focusing on specific patient sub-groups with unmet needs can unlock new growth potentials.
Leading Players in the North America Cardiac Rhythm Monitoring Industry Market
Schiller AG Biotronik SE & Co KG Shenzhen Mindray Biomedical Electronics Co Ltd Abiomed Abbott Laboratories Medtronic PLC Zoll Medical Corporation LivaNova PLC Koninklijke Philips NV Boston Scientific Corporation
Key Developments in North America Cardiac Rhythm Monitoring Industry Industry
- November 2022: Boston Scientific launched the LUX-Dx Insertable Cardiac Monitor (ICM) System, a long-term diagnostic device inserted under the skin of patients to detect arrhythmias associated with conditions such as atrial fibrillation (AF), cryptogenic stroke, and syncope in Europe.
- October 2022: Medtronic PLC received United States FDA approval for extended labeling of a cardiac lead that uses the heart's natural electrical system to deliver therapy to patients while avoiding complications.
Strategic Outlook for North America Cardiac Rhythm Monitoring Industry Market
The strategic outlook for the North America Cardiac Rhythm Monitoring Industry is exceptionally positive, driven by sustained demand from an aging demographic and continuous technological advancements. Key growth catalysts include the increasing adoption of AI-powered diagnostics for predictive and personalized patient care, the expansion of remote patient monitoring solutions facilitated by improved connectivity, and the development of more sophisticated and less invasive implantable devices. Strategic partnerships, collaborations, and targeted acquisitions will likely shape the competitive landscape, allowing companies to expand their technological portfolios and market reach. The focus on value-based healthcare and the reduction of hospital readmissions will further incentivize the adoption of advanced monitoring technologies. The industry is poised for significant innovation and growth, transforming how cardiac arrhythmias are detected, managed, and treated.
North America Cardiac Rhythm Monitoring Industry Segmentation
-
1. Product
-
1.1. Defibrillators
- 1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 1.1.2. External Defibrillators (ED)
-
1.2. Pacemakers
- 1.2.1. Implantable Pacemakers (ICPS)
- 1.2.2. External Pacemakers
-
1.3. Cardiac Resynchronization Therapy Devices
- 1.3.1. Cardiac
- 1.3.2. Cardiac
-
1.1. Defibrillators
-
2. Geography
- 2.1. United States
- 2.2. Canada
- 2.3. Mexico
North America Cardiac Rhythm Monitoring Industry Segmentation By Geography
- 1. United States
- 2. Canada
- 3. Mexico
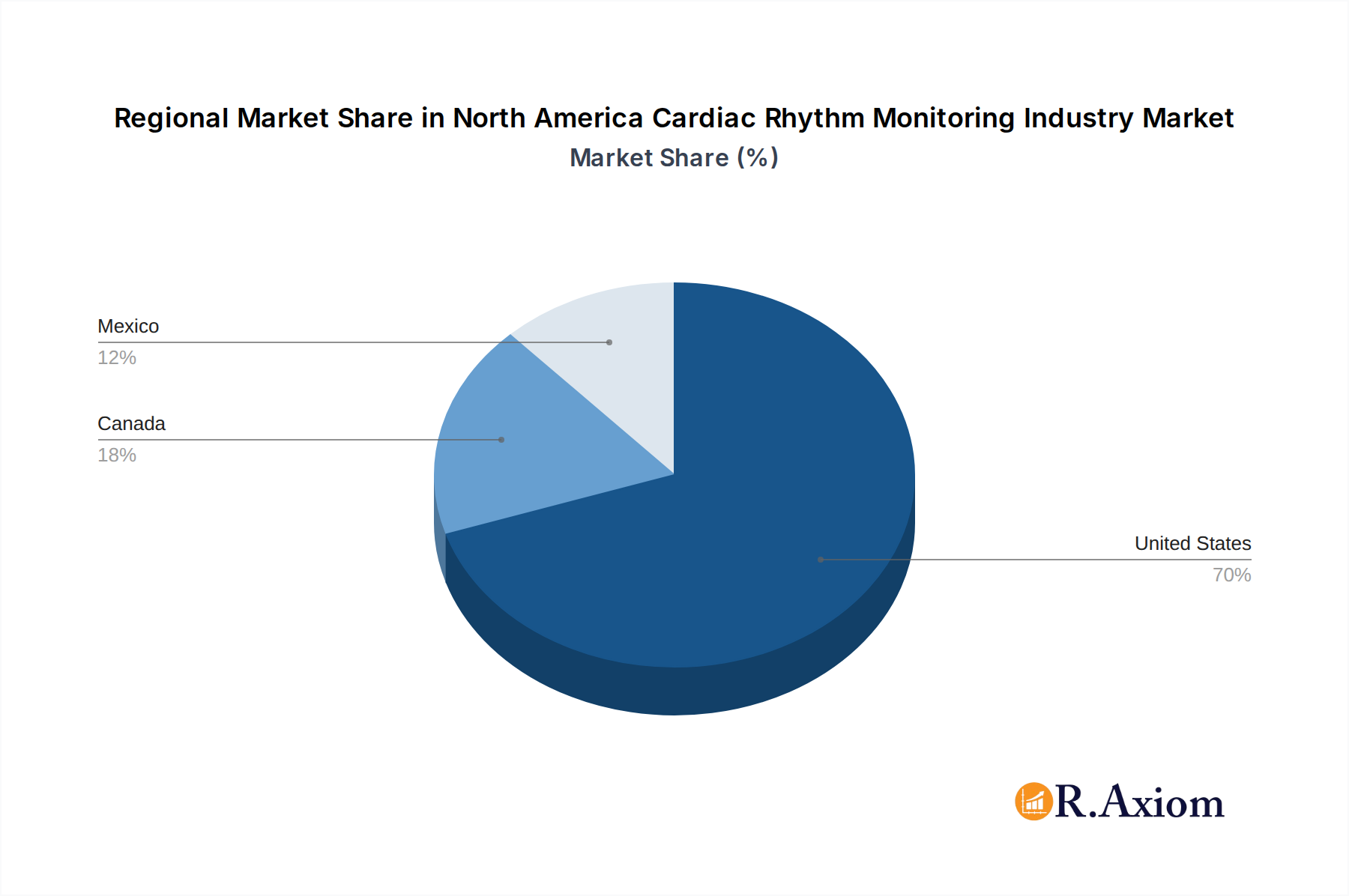
North America Cardiac Rhythm Monitoring Industry Regional Market Share

Geographic Coverage of North America Cardiac Rhythm Monitoring Industry
North America Cardiac Rhythm Monitoring Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.55% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. RAX Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product
- 5.1.1. Defibrillators
- 5.1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 5.1.1.2. External Defibrillators (ED)
- 5.1.2. Pacemakers
- 5.1.2.1. Implantable Pacemakers (ICPS)
- 5.1.2.2. External Pacemakers
- 5.1.3. Cardiac Resynchronization Therapy Devices
- 5.1.3.1. Cardiac
- 5.1.3.2. Cardiac
- 5.1.1. Defibrillators
- 5.2. Market Analysis, Insights and Forecast - by Geography
- 5.2.1. United States
- 5.2.2. Canada
- 5.2.3. Mexico
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. United States
- 5.3.2. Canada
- 5.3.3. Mexico
- 5.1. Market Analysis, Insights and Forecast - by Product
- 6. North America Cardiac Rhythm Monitoring Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product
- 6.1.1. Defibrillators
- 6.1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 6.1.1.2. External Defibrillators (ED)
- 6.1.2. Pacemakers
- 6.1.2.1. Implantable Pacemakers (ICPS)
- 6.1.2.2. External Pacemakers
- 6.1.3. Cardiac Resynchronization Therapy Devices
- 6.1.3.1. Cardiac
- 6.1.3.2. Cardiac
- 6.1.1. Defibrillators
- 6.2. Market Analysis, Insights and Forecast - by Geography
- 6.2.1. United States
- 6.2.2. Canada
- 6.2.3. Mexico
- 6.1. Market Analysis, Insights and Forecast - by Product
- 7. United States North America Cardiac Rhythm Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Product
- 7.1.1. Defibrillators
- 7.1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 7.1.1.2. External Defibrillators (ED)
- 7.1.2. Pacemakers
- 7.1.2.1. Implantable Pacemakers (ICPS)
- 7.1.2.2. External Pacemakers
- 7.1.3. Cardiac Resynchronization Therapy Devices
- 7.1.3.1. Cardiac
- 7.1.3.2. Cardiac
- 7.1.1. Defibrillators
- 7.2. Market Analysis, Insights and Forecast - by Geography
- 7.2.1. United States
- 7.2.2. Canada
- 7.2.3. Mexico
- 7.1. Market Analysis, Insights and Forecast - by Product
- 8. Canada North America Cardiac Rhythm Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Product
- 8.1.1. Defibrillators
- 8.1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 8.1.1.2. External Defibrillators (ED)
- 8.1.2. Pacemakers
- 8.1.2.1. Implantable Pacemakers (ICPS)
- 8.1.2.2. External Pacemakers
- 8.1.3. Cardiac Resynchronization Therapy Devices
- 8.1.3.1. Cardiac
- 8.1.3.2. Cardiac
- 8.1.1. Defibrillators
- 8.2. Market Analysis, Insights and Forecast - by Geography
- 8.2.1. United States
- 8.2.2. Canada
- 8.2.3. Mexico
- 8.1. Market Analysis, Insights and Forecast - by Product
- 9. Mexico North America Cardiac Rhythm Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Product
- 9.1.1. Defibrillators
- 9.1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 9.1.1.2. External Defibrillators (ED)
- 9.1.2. Pacemakers
- 9.1.2.1. Implantable Pacemakers (ICPS)
- 9.1.2.2. External Pacemakers
- 9.1.3. Cardiac Resynchronization Therapy Devices
- 9.1.3.1. Cardiac
- 9.1.3.2. Cardiac
- 9.1.1. Defibrillators
- 9.2. Market Analysis, Insights and Forecast - by Geography
- 9.2.1. United States
- 9.2.2. Canada
- 9.2.3. Mexico
- 9.1. Market Analysis, Insights and Forecast - by Product
- 10. Competitive Analysis
- 10.1. Company Profiles
- 10.1.1 SchillerAG
- 10.1.1.1. Company Overview
- 10.1.1.2. Products
- 10.1.1.3. Company Financials
- 10.1.1.4. SWOT Analysis
- 10.1.2 Biotronik SE & Co KG
- 10.1.2.1. Company Overview
- 10.1.2.2. Products
- 10.1.2.3. Company Financials
- 10.1.2.4. SWOT Analysis
- 10.1.3 Shenzhen Mindray Biomedical Electronics Co Ltd
- 10.1.3.1. Company Overview
- 10.1.3.2. Products
- 10.1.3.3. Company Financials
- 10.1.3.4. SWOT Analysis
- 10.1.4 Abiomed
- 10.1.4.1. Company Overview
- 10.1.4.2. Products
- 10.1.4.3. Company Financials
- 10.1.4.4. SWOT Analysis
- 10.1.5 Abbott Laboratories
- 10.1.5.1. Company Overview
- 10.1.5.2. Products
- 10.1.5.3. Company Financials
- 10.1.5.4. SWOT Analysis
- 10.1.6 Medtronic PLC
- 10.1.6.1. Company Overview
- 10.1.6.2. Products
- 10.1.6.3. Company Financials
- 10.1.6.4. SWOT Analysis
- 10.1.7 Zoll Medical Corporation
- 10.1.7.1. Company Overview
- 10.1.7.2. Products
- 10.1.7.3. Company Financials
- 10.1.7.4. SWOT Analysis
- 10.1.8 LivaNova PLC
- 10.1.8.1. Company Overview
- 10.1.8.2. Products
- 10.1.8.3. Company Financials
- 10.1.8.4. SWOT Analysis
- 10.1.9 Koninklijke Philips NV
- 10.1.9.1. Company Overview
- 10.1.9.2. Products
- 10.1.9.3. Company Financials
- 10.1.9.4. SWOT Analysis
- 10.1.10 Boston Scientific Corporation
- 10.1.10.1. Company Overview
- 10.1.10.2. Products
- 10.1.10.3. Company Financials
- 10.1.10.4. SWOT Analysis
- 10.1.1 SchillerAG
- 10.2. Market Entropy
- 10.2.1 Company's Key Areas Served
- 10.2.2 Recent Developments
- 10.3. Company Market Share Analysis 2025
- 10.3.1 Top 5 Companies Market Share Analysis
- 10.3.2 Top 3 Companies Market Share Analysis
- 10.4. List of Potential Customers
- 11. Research Methodology
List of Figures
- Figure 1: North America Cardiac Rhythm Monitoring Industry Revenue Breakdown (undefined, %) by Product 2025 & 2033
- Figure 2: North America Cardiac Rhythm Monitoring Industry Share (%) by Company 2025
List of Tables
- Table 1: North America Cardiac Rhythm Monitoring Industry Revenue undefined Forecast, by Product 2020 & 2033
- Table 2: North America Cardiac Rhythm Monitoring Industry Volume K Units Forecast, by Product 2020 & 2033
- Table 3: North America Cardiac Rhythm Monitoring Industry Revenue undefined Forecast, by Geography 2020 & 2033
- Table 4: North America Cardiac Rhythm Monitoring Industry Volume K Units Forecast, by Geography 2020 & 2033
- Table 5: North America Cardiac Rhythm Monitoring Industry Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: North America Cardiac Rhythm Monitoring Industry Volume K Units Forecast, by Region 2020 & 2033
- Table 7: North America Cardiac Rhythm Monitoring Industry Revenue undefined Forecast, by Product 2020 & 2033
- Table 8: North America Cardiac Rhythm Monitoring Industry Volume K Units Forecast, by Product 2020 & 2033
- Table 9: North America Cardiac Rhythm Monitoring Industry Revenue undefined Forecast, by Geography 2020 & 2033
- Table 10: North America Cardiac Rhythm Monitoring Industry Volume K Units Forecast, by Geography 2020 & 2033
- Table 11: North America Cardiac Rhythm Monitoring Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 12: North America Cardiac Rhythm Monitoring Industry Volume K Units Forecast, by Country 2020 & 2033
- Table 13: North America Cardiac Rhythm Monitoring Industry Revenue undefined Forecast, by Product 2020 & 2033
- Table 14: North America Cardiac Rhythm Monitoring Industry Volume K Units Forecast, by Product 2020 & 2033
- Table 15: North America Cardiac Rhythm Monitoring Industry Revenue undefined Forecast, by Geography 2020 & 2033
- Table 16: North America Cardiac Rhythm Monitoring Industry Volume K Units Forecast, by Geography 2020 & 2033
- Table 17: North America Cardiac Rhythm Monitoring Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 18: North America Cardiac Rhythm Monitoring Industry Volume K Units Forecast, by Country 2020 & 2033
- Table 19: North America Cardiac Rhythm Monitoring Industry Revenue undefined Forecast, by Product 2020 & 2033
- Table 20: North America Cardiac Rhythm Monitoring Industry Volume K Units Forecast, by Product 2020 & 2033
- Table 21: North America Cardiac Rhythm Monitoring Industry Revenue undefined Forecast, by Geography 2020 & 2033
- Table 22: North America Cardiac Rhythm Monitoring Industry Volume K Units Forecast, by Geography 2020 & 2033
- Table 23: North America Cardiac Rhythm Monitoring Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 24: North America Cardiac Rhythm Monitoring Industry Volume K Units Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the North America Cardiac Rhythm Monitoring Industry?
The projected CAGR is approximately 6.55%.
2. Which companies are prominent players in the North America Cardiac Rhythm Monitoring Industry?
Key companies in the market include SchillerAG, Biotronik SE & Co KG, Shenzhen Mindray Biomedical Electronics Co Ltd, Abiomed, Abbott Laboratories, Medtronic PLC, Zoll Medical Corporation, LivaNova PLC, Koninklijke Philips NV, Boston Scientific Corporation.
3. What are the main segments of the North America Cardiac Rhythm Monitoring Industry?
The market segments include Product, Geography.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
CVDs. including arrhythmias. heart failure. and ischemic heart disease. are a major cause of morbidity and mortality in North America. The aging population. rising incidence of hypertension. diabetes. and obesity. along with unhealthy lifestyles. are contributing factors to the high prevalence of CVDs..
6. What are the notable trends driving market growth?
AI and machine learning (ML) technologies are being increasingly integrated into CRM devices. allowing for better automated detection of abnormal heart rhythms and reducing false alarms. These technologies also enable more accurate predictive analytics for preventing cardiac events. such as heart attacks and strokes..
7. Are there any restraints impacting market growth?
Despite advancements in technology. many CRM devices are still expensive. particularly implantable devices like ICDs and CRTs. High costs can limit patient access. especially among those who lack adequate insurance coverage or live in regions with restricted healthcare budgets..
8. Can you provide examples of recent developments in the market?
Nov 2022: Boston Scientific the LUX-Dx Insertable Cardiac Monitor (ICM) System, a long-term diagnostic device inserted under the skin of patients to detect arrhythmias associated with conditions such as atrial fibrillation (AF), cryptogenic stroke, and syncope in Europe.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in K Units.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "North America Cardiac Rhythm Monitoring Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the North America Cardiac Rhythm Monitoring Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the North America Cardiac Rhythm Monitoring Industry?
To stay informed about further developments, trends, and reports in the North America Cardiac Rhythm Monitoring Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence