Key Insights
The global Pituitary Cancer Diagnostics market is poised for significant expansion, projected to reach an estimated $2.27 million by 2025, driven by a robust Compound Annual Growth Rate (CAGR) of 8.30% throughout the forecast period of 2025-2033. This growth is primarily fueled by advancements in diagnostic technologies, increasing awareness of pituitary disorders, and a rising incidence of pituitary tumors, both primary and metastatic. Early and accurate diagnosis is paramount for effective treatment and improved patient outcomes, propelling the demand for sophisticated diagnostic tools and services. The market is segmented across various diagnostic types, including advanced imaging techniques like MRI and CT scans, alongside molecular testing and biopsies, each playing a crucial role in the comprehensive evaluation of pituitary cancers. The increasing prevalence of challenging tumor types such as Glioblastoma and Meningioma further stimulates innovation and market penetration for specialized diagnostic solutions.
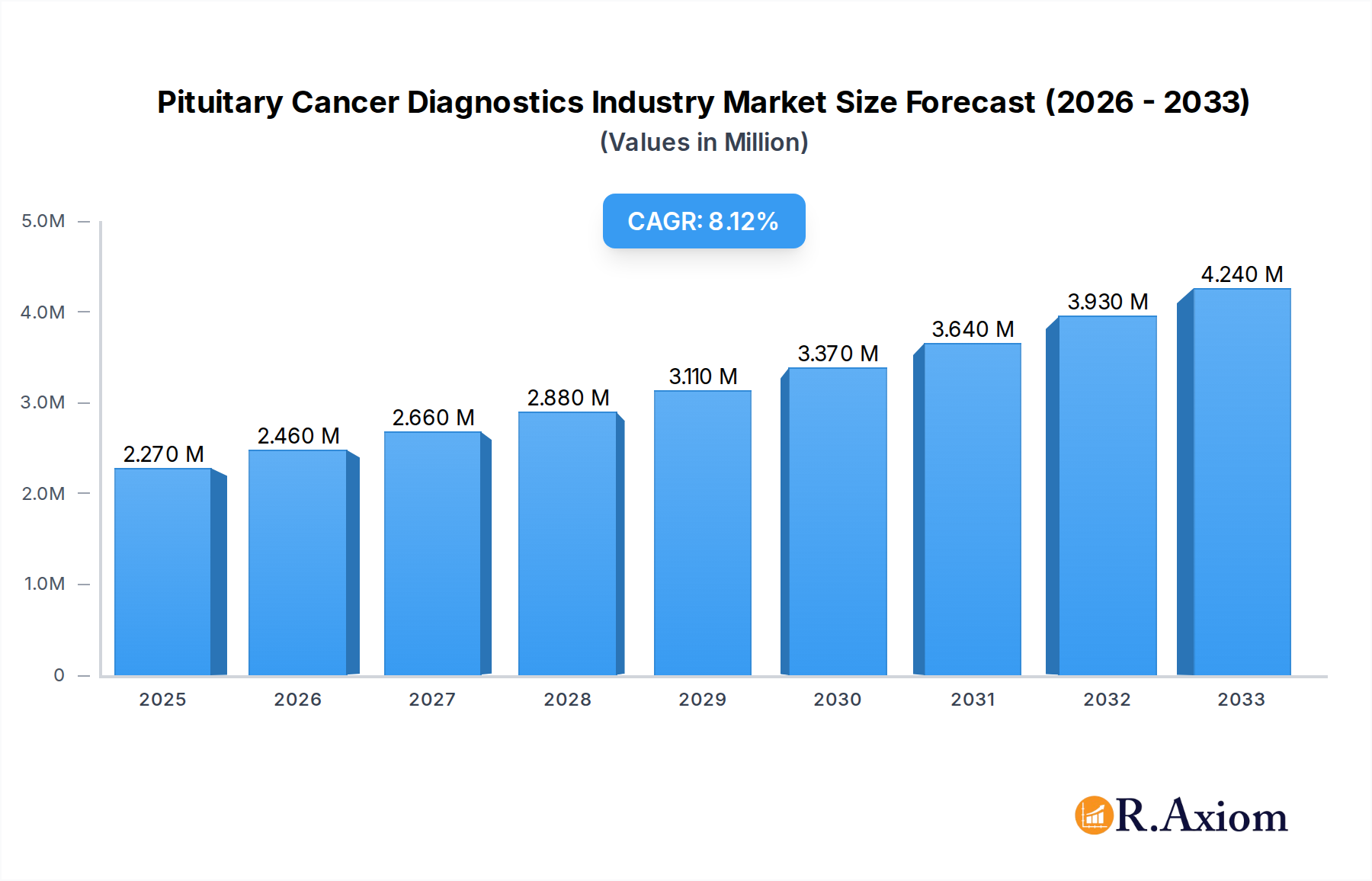
Pituitary Cancer Diagnostics Industry Market Size (In Million)

The competitive landscape features key players like Siemens Healthineers, GE Healthcare, and Thermo Fisher Scientific, actively investing in research and development to enhance diagnostic accuracy and accessibility. The demand for these diagnostic services is predominantly from hospitals and dedicated diagnostic centers, reflecting the need for specialized equipment and expertise. Geographically, North America and Europe are expected to lead the market due to established healthcare infrastructures, high healthcare expenditure, and a strong emphasis on early disease detection. However, the Asia Pacific region presents a substantial growth opportunity, driven by improving healthcare facilities, a growing patient pool, and increasing government initiatives to strengthen diagnostic capabilities. While market growth is robust, potential restraints could include the high cost of advanced diagnostic equipment and the need for skilled professionals to interpret complex diagnostic data, though these are expected to be mitigated by technological advancements and increasing market demand.
Pituitary Cancer Diagnostics Industry Company Market Share

This in-depth market research report provides an exhaustive analysis of the global Pituitary Cancer Diagnostics Industry. Covering the historical period from 2019 to 2024, the base year 2025, and a comprehensive forecast period extending to 2033, this report is your definitive guide to understanding market dynamics, key players, emerging trends, and future growth catalysts. We delve into the intricate segmentation of the market by diagnostic type, tumor type, and end-user, offering actionable insights for stakeholders across the healthcare and diagnostics ecosystem. Our analysis is driven by high-traffic keywords essential for search engine optimization and engaging industry professionals seeking critical market intelligence.
Pituitary Cancer Diagnostics Industry Market Concentration & Innovation
The Pituitary Cancer Diagnostics Industry exhibits a moderate level of market concentration, with key players investing heavily in research and development to drive innovation. Factors such as advancements in molecular diagnostics and liquid biopsy technologies are significantly influencing product development and market entry strategies. Regulatory frameworks, while crucial for patient safety, can also present hurdles for novel diagnostic solutions. The presence of potent product substitutes, such as advanced imaging techniques and existing diagnostic protocols, necessitates continuous innovation to maintain competitive advantage. End-user preferences are increasingly shifting towards less invasive and more accurate diagnostic methods, particularly molecular testing. Mergers and Acquisitions (M&A) activities are expected to shape the market landscape, with strategic alliances aimed at consolidating market share and expanding technological capabilities. Estimated M&A deal values are projected to reach xx Million in the forecast period. Leading companies are focusing on expanding their product portfolios and geographical reach through strategic partnerships and acquisitions.
- Market Share Snapshot: Key players like Siemens Healthineers and GE Healthcare hold significant market shares, driven by their extensive product offerings and established distribution networks.
- Innovation Drivers: Growing demand for early and precise detection of pituitary tumors, coupled with technological breakthroughs in imaging and genetic sequencing, are primary innovation catalysts.
- Regulatory Landscape: Stringent regulatory approvals, such as FDA and EMA certifications, are essential but can impact the speed of new product introductions.
- M&A Activity: Companies are actively pursuing M&A to acquire innovative technologies, expand their market presence, and gain a competitive edge.
Pituitary Cancer Diagnostics Industry Industry Trends & Insights
The global Pituitary Cancer Diagnostics Industry is poised for robust growth, driven by a confluence of factors including increasing cancer incidence, advancements in diagnostic technologies, and rising healthcare expenditure worldwide. The market's compound annual growth rate (CAGR) is estimated to be xx% during the forecast period (2025-2033). A significant trend is the growing adoption of advanced imaging techniques such as Magnetic Resonance Imaging (MRI) and Computed Tomography (CT) scans, which offer superior resolution and detail in visualizing pituitary abnormalities. Furthermore, the burgeoning field of molecular diagnostics, including genetic testing and biomarker analysis, is revolutionizing early detection and personalized treatment strategies for pituitary cancers. The rising prevalence of endocrine disorders and hormonal imbalances, often indicative of pituitary abnormalities, also contributes to market expansion. Patient awareness regarding early diagnosis and treatment options is also on an upward trajectory, fueling demand for sophisticated diagnostic solutions. The competitive landscape is characterized by intense R&D investments, strategic collaborations, and a focus on developing minimally invasive diagnostic methods. The integration of artificial intelligence (AI) and machine learning (ML) in image analysis and data interpretation is emerging as a key technological disruption, promising enhanced accuracy and efficiency. Consumer preferences are leaning towards diagnostic services that offer speed, accuracy, and a less invasive patient experience. This is evident in the growing interest in liquid biopsy techniques for non-invasive tumor detection. The penetration of advanced diagnostic tools is increasing across both developed and developing economies, albeit at varying paces. The global market size is projected to reach $xx Million by 2033.
Dominant Markets & Segments in Pituitary Cancer Diagnostics Industry
The Imaging Test segment, encompassing MRI, CT Scan, and other advanced imaging modalities, currently dominates the Pituitary Cancer Diagnostics Industry. This dominance is attributed to the established efficacy and widespread availability of these techniques in visualizing pituitary gland abnormalities and associated tumors.
Diagnostic Type Dominance:
- Imaging Test (MRI, CT Scan, Other): Remains the primary diagnostic modality due to its non-invasive nature, high sensitivity in detecting structural changes, and widespread adoption in clinical practice. The increasing resolution and advanced functionalities of MRI and CT scanners further solidify its leading position.
- Molecular Testing: Emerging as a significant growth segment, driven by the need for precise tumor subtyping, identification of actionable mutations, and prediction of treatment response.
- Biopsy: While invasive, it remains crucial for definitive histological diagnosis and grading of pituitary tumors.
- Lumbar Puncture: Used in specific cases for cerebrospinal fluid analysis to detect tumor spread or inflammation.
Tumor Type Dominance:
- Pituitary Endoma: As the most common type of pituitary tumor, it naturally drives the demand for diagnostics.
- Meningioma: While not exclusively pituitary, meningiomas in the vicinity of the pituitary gland require sophisticated imaging for diagnosis.
- Metastatic and Glioblastoma: Though rarer, the accurate diagnosis of these aggressive tumor types is critical and relies on advanced diagnostic tools.
End User Dominance:
- Hospitals: Constitute the largest end-user segment due to their comprehensive diagnostic infrastructure, specialized medical staff, and patient volume. They are at the forefront of adopting new diagnostic technologies and performing complex procedures.
- Diagnostics Centers: Increasingly playing a vital role, particularly in providing specialized imaging and molecular testing services, catering to outpatient needs and referring physicians.
- Others (Research Institutions, Specialty Clinics): Contribute to market growth through specialized research and niche diagnostic services.
The strong performance of hospitals is bolstered by government healthcare initiatives, increasing healthcare spending, and the presence of advanced medical facilities. Economic policies promoting healthcare infrastructure development and technological adoption further support the growth of these dominant segments.
Pituitary Cancer Diagnostics Industry Product Developments
The Pituitary Cancer Diagnostics Industry is witnessing a surge in product innovations aimed at enhancing diagnostic accuracy, reducing invasiveness, and enabling personalized treatment. Advancements in molecular diagnostics, including next-generation sequencing (NGS) and ctDNA analysis, are paving the way for early detection and precise tumor profiling. Novel antibody development, as seen with Roche's recent launches, is crucial for identifying specific mutations in brain cancers, indirectly benefiting pituitary tumor diagnostics. Liquid biopsy technologies are gaining traction, offering a less invasive alternative to traditional biopsies for detecting tumor DNA in blood. AI-powered image analysis software is also emerging, promising to improve the interpretation of MRI and CT scans, leading to faster and more accurate diagnoses. These developments offer competitive advantages by providing clinicians with more precise diagnostic information, leading to better patient management and outcomes.
Report Scope & Segmentation Analysis
This report comprehensively analyzes the Pituitary Cancer Diagnostics Industry across its key segments.
- Diagnostic Type: Includes Imaging Tests (MRI, CT Scan, Other), Lumbar Puncture, Biopsy, Molecular Testing, and Others. The Imaging Test segment is projected to hold the largest market share, with Molecular Testing exhibiting the highest growth rate due to technological advancements.
- Tumor Type: Encompasses Metastatic, Meningioma, Glioblastoma, Pituitary Endoma, and Others. Pituitary Endoma is expected to remain the most prevalent segment driving demand.
- End User: Covers Hospitals, Diagnostics Centers, and Others. Hospitals will continue to dominate the market due to extensive diagnostic capabilities and patient influx.
Key Drivers of Pituitary Cancer Diagnostics Industry Growth
The Pituitary Cancer Diagnostics Industry's growth is propelled by several key factors.
- Technological Advancements: Innovations in imaging technology (e.g., higher resolution MRI, AI-assisted CT) and molecular diagnostics (e.g., liquid biopsy, genetic sequencing) are crucial.
- Increasing Cancer Incidence and Awareness: A rising global incidence of pituitary tumors and growing public awareness about early detection and treatment options are significant drivers.
- Rising Healthcare Expenditure: Increased investment in healthcare infrastructure and advanced medical technologies, particularly in emerging economies, fuels market expansion.
- Advancements in Treatment Modalities: The development of targeted therapies necessitates precise diagnostic tools for patient stratification.
Challenges in the Pituitary Cancer Diagnostics Industry Sector
Despite promising growth, the sector faces several challenges.
- High Cost of Advanced Diagnostics: The expense associated with sophisticated imaging equipment and molecular testing can be a barrier for some healthcare providers and patients.
- Regulatory Hurdles: Obtaining approvals for new diagnostic technologies can be a lengthy and complex process, potentially delaying market entry.
- Shortage of Skilled Professionals: A lack of trained personnel to operate advanced diagnostic equipment and interpret complex molecular data can hinder adoption.
- Reimbursement Policies: Inconsistent or inadequate reimbursement policies for novel diagnostic procedures can impact their widespread use.
Emerging Opportunities in Pituitary Cancer Diagnostics Industry
The Pituitary Cancer Diagnostics Industry is ripe with emerging opportunities.
- Growth of Liquid Biopsy: The development and validation of non-invasive liquid biopsy tests present a significant opportunity for early detection and monitoring.
- AI and Machine Learning Integration: Leveraging AI for image analysis and data interpretation can enhance diagnostic accuracy and efficiency.
- Personalized Medicine: The growing focus on personalized medicine drives demand for genetic and molecular profiling of tumors to tailor treatment strategies.
- Emerging Markets: Untapped potential in developing economies, with increasing healthcare investments, offers substantial growth avenues.
Leading Players in the Pituitary Cancer Diagnostics Industry Market
- Siemens Healthineers
- Hologic Inc
- GE Healthcare
- Hitachi Ltd
- Koninklijke Philips N V
- Fujifilm Corporation
- Thermo Fisher Scientific
- NantOmics
Key Developments in Pituitary Cancer Diagnostics Industry Industry
- February 2023: Roche announced the launch of the IDH1 R132H (MRQ-67) Rabbit Monoclonal Primary Antibody and the ATRX Rabbit Polyclonal Antibody to identify mutation status in patients diagnosed with brain cancer.
- January 2023: Datar Cancer Genetics Inc. received FDA breakthrough device designation for the TriNetra-Glio blood test to help in the diagnosis of brain tumors.
Strategic Outlook for Pituitary Cancer Diagnostics Industry Market
The strategic outlook for the Pituitary Cancer Diagnostics Industry is highly optimistic, driven by relentless innovation and increasing global demand for accurate and early tumor detection. Key growth catalysts include the expanding application of artificial intelligence in diagnostic imaging, the burgeoning field of liquid biopsy for non-invasive diagnostics, and the continuous development of targeted therapies that necessitate precise molecular profiling. Companies that invest in R&D, forge strategic partnerships, and focus on developing cost-effective and accessible diagnostic solutions will be well-positioned to capitalize on the significant future market potential. The emphasis on personalized medicine will further propel the adoption of advanced genomic and proteomic diagnostic tools, creating a dynamic and evolving market landscape.
Pituitary Cancer Diagnostics Industry Segmentation
-
1. Diagnostic Type
-
1.1. Imaging Test
- 1.1.1. MRI
- 1.1.2. CT Scan
- 1.1.3. Other
- 1.2. Lumbar Puncture
- 1.3. Biopsy
- 1.4. Molecular Testing
- 1.5. Others
-
1.1. Imaging Test
-
2. Tumor Type
- 2.1. Metastatic
- 2.2. Meningioma
- 2.3. Glioblastoma
- 2.4. Pituitary Endoma
- 2.5. Others
-
3. End User
- 3.1. Hospitals
- 3.2. Diagnostics Centers
- 3.3. Others
Pituitary Cancer Diagnostics Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
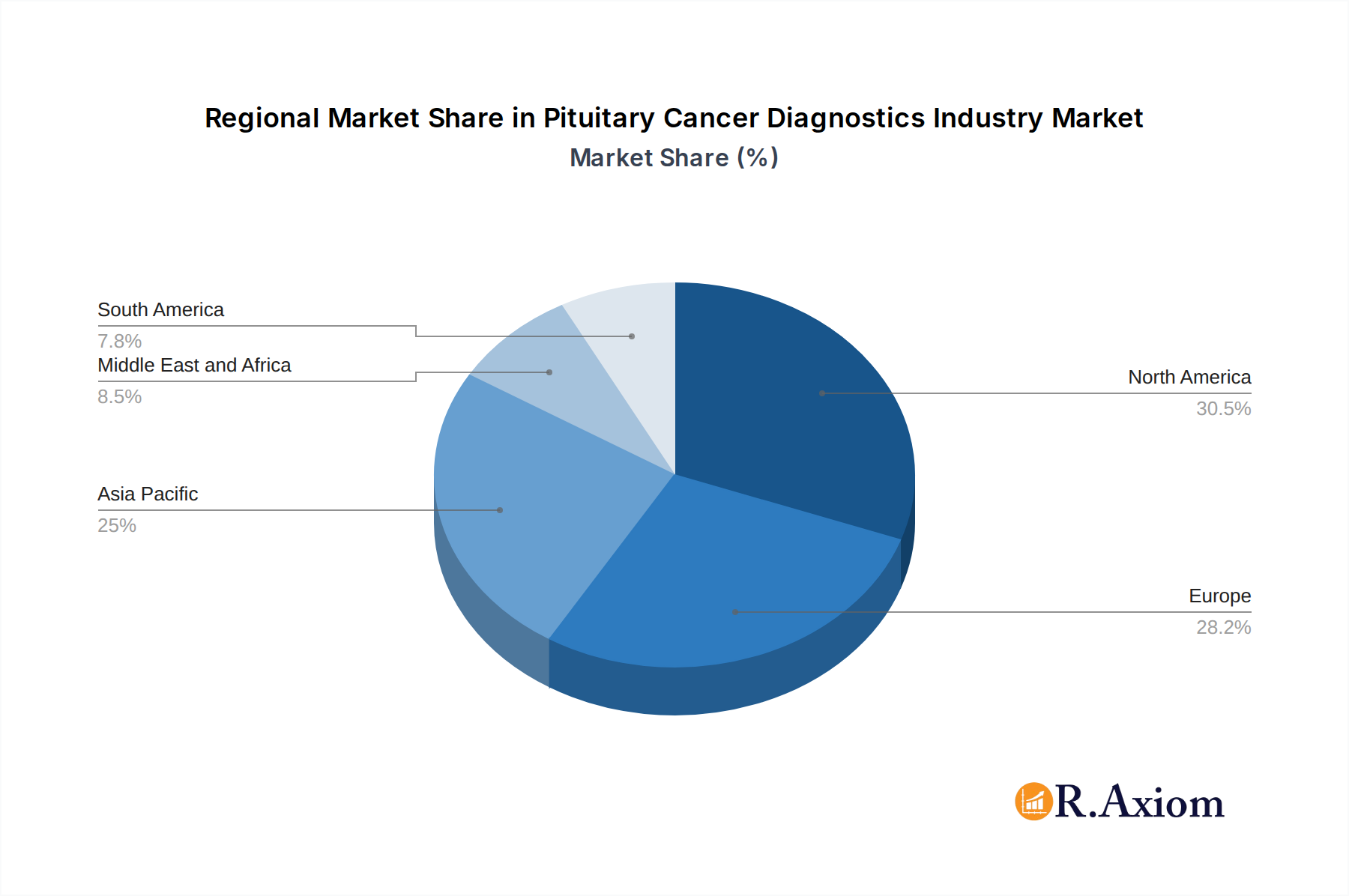
Pituitary Cancer Diagnostics Industry Regional Market Share

Geographic Coverage of Pituitary Cancer Diagnostics Industry
Pituitary Cancer Diagnostics Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.30% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. RAX Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 5.1.1. Imaging Test
- 5.1.1.1. MRI
- 5.1.1.2. CT Scan
- 5.1.1.3. Other
- 5.1.2. Lumbar Puncture
- 5.1.3. Biopsy
- 5.1.4. Molecular Testing
- 5.1.5. Others
- 5.1.1. Imaging Test
- 5.2. Market Analysis, Insights and Forecast - by Tumor Type
- 5.2.1. Metastatic
- 5.2.2. Meningioma
- 5.2.3. Glioblastoma
- 5.2.4. Pituitary Endoma
- 5.2.5. Others
- 5.3. Market Analysis, Insights and Forecast - by End User
- 5.3.1. Hospitals
- 5.3.2. Diagnostics Centers
- 5.3.3. Others
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. North America
- 5.4.2. Europe
- 5.4.3. Asia Pacific
- 5.4.4. Middle East and Africa
- 5.4.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 6. Global Pituitary Cancer Diagnostics Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 6.1.1. Imaging Test
- 6.1.1.1. MRI
- 6.1.1.2. CT Scan
- 6.1.1.3. Other
- 6.1.2. Lumbar Puncture
- 6.1.3. Biopsy
- 6.1.4. Molecular Testing
- 6.1.5. Others
- 6.1.1. Imaging Test
- 6.2. Market Analysis, Insights and Forecast - by Tumor Type
- 6.2.1. Metastatic
- 6.2.2. Meningioma
- 6.2.3. Glioblastoma
- 6.2.4. Pituitary Endoma
- 6.2.5. Others
- 6.3. Market Analysis, Insights and Forecast - by End User
- 6.3.1. Hospitals
- 6.3.2. Diagnostics Centers
- 6.3.3. Others
- 6.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 7. North America Pituitary Cancer Diagnostics Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 7.1.1. Imaging Test
- 7.1.1.1. MRI
- 7.1.1.2. CT Scan
- 7.1.1.3. Other
- 7.1.2. Lumbar Puncture
- 7.1.3. Biopsy
- 7.1.4. Molecular Testing
- 7.1.5. Others
- 7.1.1. Imaging Test
- 7.2. Market Analysis, Insights and Forecast - by Tumor Type
- 7.2.1. Metastatic
- 7.2.2. Meningioma
- 7.2.3. Glioblastoma
- 7.2.4. Pituitary Endoma
- 7.2.5. Others
- 7.3. Market Analysis, Insights and Forecast - by End User
- 7.3.1. Hospitals
- 7.3.2. Diagnostics Centers
- 7.3.3. Others
- 7.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 8. Europe Pituitary Cancer Diagnostics Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 8.1.1. Imaging Test
- 8.1.1.1. MRI
- 8.1.1.2. CT Scan
- 8.1.1.3. Other
- 8.1.2. Lumbar Puncture
- 8.1.3. Biopsy
- 8.1.4. Molecular Testing
- 8.1.5. Others
- 8.1.1. Imaging Test
- 8.2. Market Analysis, Insights and Forecast - by Tumor Type
- 8.2.1. Metastatic
- 8.2.2. Meningioma
- 8.2.3. Glioblastoma
- 8.2.4. Pituitary Endoma
- 8.2.5. Others
- 8.3. Market Analysis, Insights and Forecast - by End User
- 8.3.1. Hospitals
- 8.3.2. Diagnostics Centers
- 8.3.3. Others
- 8.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 9. Asia Pacific Pituitary Cancer Diagnostics Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 9.1.1. Imaging Test
- 9.1.1.1. MRI
- 9.1.1.2. CT Scan
- 9.1.1.3. Other
- 9.1.2. Lumbar Puncture
- 9.1.3. Biopsy
- 9.1.4. Molecular Testing
- 9.1.5. Others
- 9.1.1. Imaging Test
- 9.2. Market Analysis, Insights and Forecast - by Tumor Type
- 9.2.1. Metastatic
- 9.2.2. Meningioma
- 9.2.3. Glioblastoma
- 9.2.4. Pituitary Endoma
- 9.2.5. Others
- 9.3. Market Analysis, Insights and Forecast - by End User
- 9.3.1. Hospitals
- 9.3.2. Diagnostics Centers
- 9.3.3. Others
- 9.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 10. Middle East and Africa Pituitary Cancer Diagnostics Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 10.1.1. Imaging Test
- 10.1.1.1. MRI
- 10.1.1.2. CT Scan
- 10.1.1.3. Other
- 10.1.2. Lumbar Puncture
- 10.1.3. Biopsy
- 10.1.4. Molecular Testing
- 10.1.5. Others
- 10.1.1. Imaging Test
- 10.2. Market Analysis, Insights and Forecast - by Tumor Type
- 10.2.1. Metastatic
- 10.2.2. Meningioma
- 10.2.3. Glioblastoma
- 10.2.4. Pituitary Endoma
- 10.2.5. Others
- 10.3. Market Analysis, Insights and Forecast - by End User
- 10.3.1. Hospitals
- 10.3.2. Diagnostics Centers
- 10.3.3. Others
- 10.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 11. South America Pituitary Cancer Diagnostics Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 11.1.1. Imaging Test
- 11.1.1.1. MRI
- 11.1.1.2. CT Scan
- 11.1.1.3. Other
- 11.1.2. Lumbar Puncture
- 11.1.3. Biopsy
- 11.1.4. Molecular Testing
- 11.1.5. Others
- 11.1.1. Imaging Test
- 11.2. Market Analysis, Insights and Forecast - by Tumor Type
- 11.2.1. Metastatic
- 11.2.2. Meningioma
- 11.2.3. Glioblastoma
- 11.2.4. Pituitary Endoma
- 11.2.5. Others
- 11.3. Market Analysis, Insights and Forecast - by End User
- 11.3.1. Hospitals
- 11.3.2. Diagnostics Centers
- 11.3.3. Others
- 11.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Siemens Healthineers
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Hologic Inc
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 GE Healthcare
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Hitachi Ltd
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Koninklijke Philips N V
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Fujifilm Corporation
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Thermo Fisher Scientific
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 NantOmics
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.1 Siemens Healthineers
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Pituitary Cancer Diagnostics Industry Revenue Breakdown (Million, %) by Region 2025 & 2033
- Figure 2: North America Pituitary Cancer Diagnostics Industry Revenue (Million), by Diagnostic Type 2025 & 2033
- Figure 3: North America Pituitary Cancer Diagnostics Industry Revenue Share (%), by Diagnostic Type 2025 & 2033
- Figure 4: North America Pituitary Cancer Diagnostics Industry Revenue (Million), by Tumor Type 2025 & 2033
- Figure 5: North America Pituitary Cancer Diagnostics Industry Revenue Share (%), by Tumor Type 2025 & 2033
- Figure 6: North America Pituitary Cancer Diagnostics Industry Revenue (Million), by End User 2025 & 2033
- Figure 7: North America Pituitary Cancer Diagnostics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 8: North America Pituitary Cancer Diagnostics Industry Revenue (Million), by Country 2025 & 2033
- Figure 9: North America Pituitary Cancer Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 10: Europe Pituitary Cancer Diagnostics Industry Revenue (Million), by Diagnostic Type 2025 & 2033
- Figure 11: Europe Pituitary Cancer Diagnostics Industry Revenue Share (%), by Diagnostic Type 2025 & 2033
- Figure 12: Europe Pituitary Cancer Diagnostics Industry Revenue (Million), by Tumor Type 2025 & 2033
- Figure 13: Europe Pituitary Cancer Diagnostics Industry Revenue Share (%), by Tumor Type 2025 & 2033
- Figure 14: Europe Pituitary Cancer Diagnostics Industry Revenue (Million), by End User 2025 & 2033
- Figure 15: Europe Pituitary Cancer Diagnostics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 16: Europe Pituitary Cancer Diagnostics Industry Revenue (Million), by Country 2025 & 2033
- Figure 17: Europe Pituitary Cancer Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 18: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue (Million), by Diagnostic Type 2025 & 2033
- Figure 19: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue Share (%), by Diagnostic Type 2025 & 2033
- Figure 20: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue (Million), by Tumor Type 2025 & 2033
- Figure 21: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue Share (%), by Tumor Type 2025 & 2033
- Figure 22: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue (Million), by End User 2025 & 2033
- Figure 23: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 24: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue (Million), by Country 2025 & 2033
- Figure 25: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 26: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue (Million), by Diagnostic Type 2025 & 2033
- Figure 27: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue Share (%), by Diagnostic Type 2025 & 2033
- Figure 28: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue (Million), by Tumor Type 2025 & 2033
- Figure 29: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue Share (%), by Tumor Type 2025 & 2033
- Figure 30: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue (Million), by End User 2025 & 2033
- Figure 31: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 32: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue (Million), by Country 2025 & 2033
- Figure 33: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 34: South America Pituitary Cancer Diagnostics Industry Revenue (Million), by Diagnostic Type 2025 & 2033
- Figure 35: South America Pituitary Cancer Diagnostics Industry Revenue Share (%), by Diagnostic Type 2025 & 2033
- Figure 36: South America Pituitary Cancer Diagnostics Industry Revenue (Million), by Tumor Type 2025 & 2033
- Figure 37: South America Pituitary Cancer Diagnostics Industry Revenue Share (%), by Tumor Type 2025 & 2033
- Figure 38: South America Pituitary Cancer Diagnostics Industry Revenue (Million), by End User 2025 & 2033
- Figure 39: South America Pituitary Cancer Diagnostics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 40: South America Pituitary Cancer Diagnostics Industry Revenue (Million), by Country 2025 & 2033
- Figure 41: South America Pituitary Cancer Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Diagnostic Type 2020 & 2033
- Table 2: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Tumor Type 2020 & 2033
- Table 3: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by End User 2020 & 2033
- Table 4: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Region 2020 & 2033
- Table 5: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Diagnostic Type 2020 & 2033
- Table 6: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Tumor Type 2020 & 2033
- Table 7: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by End User 2020 & 2033
- Table 8: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 9: United States Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 10: Canada Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 11: Mexico Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 12: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Diagnostic Type 2020 & 2033
- Table 13: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Tumor Type 2020 & 2033
- Table 14: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by End User 2020 & 2033
- Table 15: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 16: Germany Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 17: United Kingdom Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 18: France Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 19: Italy Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 20: Spain Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 21: Rest of Europe Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 22: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Diagnostic Type 2020 & 2033
- Table 23: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Tumor Type 2020 & 2033
- Table 24: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by End User 2020 & 2033
- Table 25: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 26: China Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 27: Japan Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 28: India Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 29: Australia Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 30: South Korea Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 31: Rest of Asia Pacific Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 32: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Diagnostic Type 2020 & 2033
- Table 33: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Tumor Type 2020 & 2033
- Table 34: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by End User 2020 & 2033
- Table 35: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 36: GCC Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 37: South Africa Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 38: Rest of Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 39: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Diagnostic Type 2020 & 2033
- Table 40: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Tumor Type 2020 & 2033
- Table 41: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by End User 2020 & 2033
- Table 42: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 43: Brazil Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 44: Argentina Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 45: Rest of South America Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Pituitary Cancer Diagnostics Industry?
The projected CAGR is approximately 8.30%.
2. Which companies are prominent players in the Pituitary Cancer Diagnostics Industry?
Key companies in the market include Siemens Healthineers, Hologic Inc, GE Healthcare, Hitachi Ltd, Koninklijke Philips N V, Fujifilm Corporation, Thermo Fisher Scientific, NantOmics.
3. What are the main segments of the Pituitary Cancer Diagnostics Industry?
The market segments include Diagnostic Type, Tumor Type, End User.
4. Can you provide details about the market size?
The market size is estimated to be USD 2.27 Million as of 2022.
5. What are some drivers contributing to market growth?
Increasing Prevalence of Neurological Disorders and Cancers; Rising Number of Smokers.
6. What are the notable trends driving market growth?
MRI segment is Expected to Hold a Significant Share in the Brain Tumor Diagnostics Market.
7. Are there any restraints impacting market growth?
High Costs Associated with Diagnosis.
8. Can you provide examples of recent developments in the market?
February 2023: Roche announced the launch of the IDH1 R132H (MRQ-67) Rabbit Monoclonal Primary Antibody and the ATRX Rabbit Polyclonal Antibody to identify mutation status in patients diagnosed with brain cancer.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Pituitary Cancer Diagnostics Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Pituitary Cancer Diagnostics Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Pituitary Cancer Diagnostics Industry?
To stay informed about further developments, trends, and reports in the Pituitary Cancer Diagnostics Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
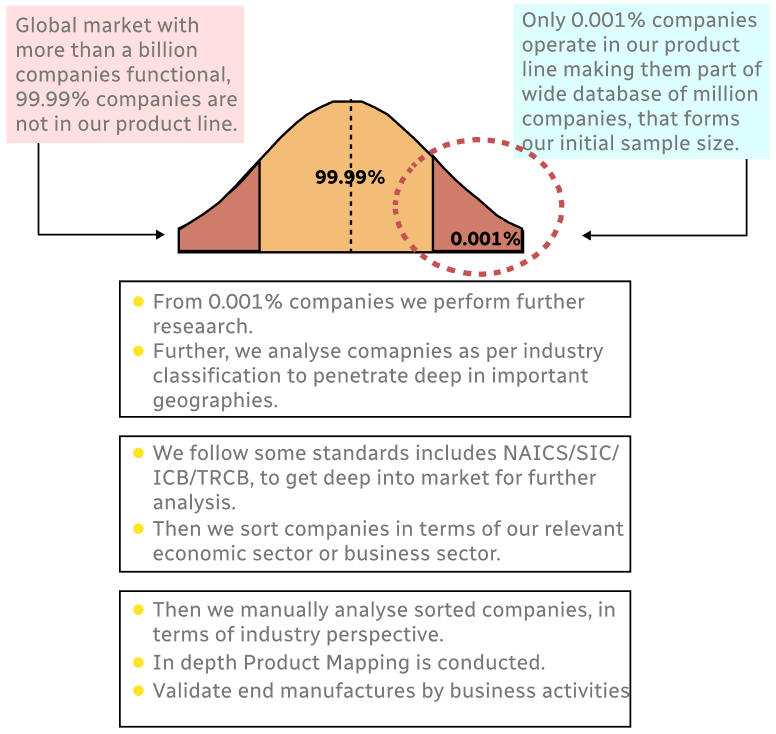
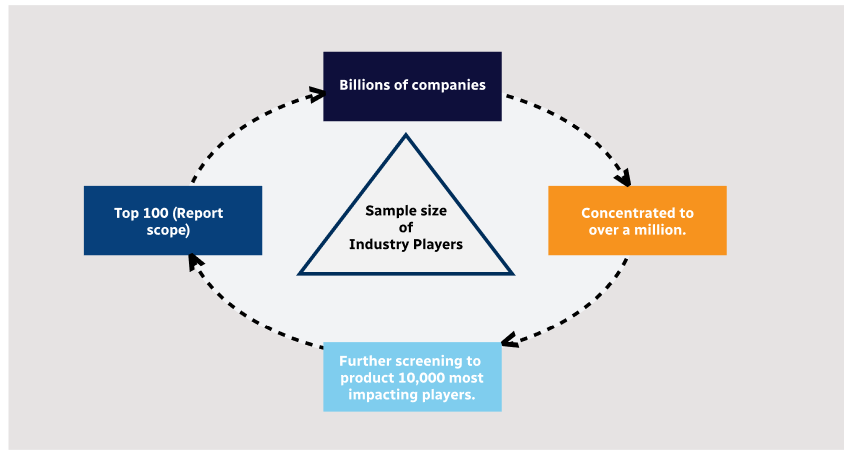
Step 1 - Identification of Relevant Samples Size from Population Database
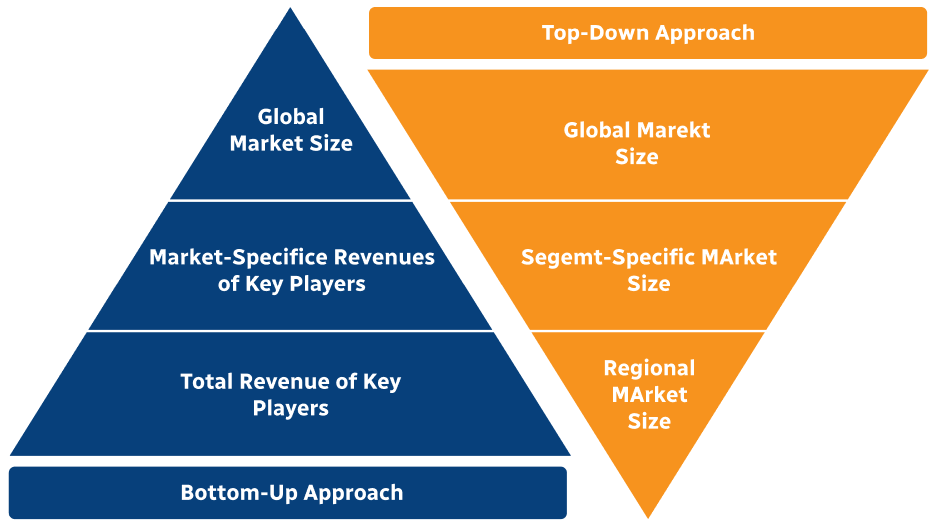
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence