Key Insights
The global veterinary ceftiofur hydrochloride injection market is experiencing robust growth, driven by the increasing prevalence of bacterial infections in livestock and companion animals, coupled with the drug's broad-spectrum efficacy and relatively low cost compared to other antibiotics. The market's Compound Annual Growth Rate (CAGR) is estimated to be around 6-8% between 2025 and 2033, indicating a significant expansion opportunity. Key drivers include rising animal healthcare expenditure globally, increased veterinary adoption of injectable antibiotics for treating severe infections, and a growing demand for efficient and effective treatments in intensive animal farming operations. Market segmentation reveals a significant share held by large-scale pharmaceutical manufacturers, with a concentration in North America and Europe, reflecting higher regulatory standards and greater veterinary penetration in these regions. However, the market faces constraints including growing concerns regarding antibiotic resistance and the stringent regulatory landscape surrounding veterinary drug approvals, which necessitates robust clinical trials and regulatory filings for market entry. The rise of alternative treatment modalities, such as phage therapy, also presents a longer-term challenge.
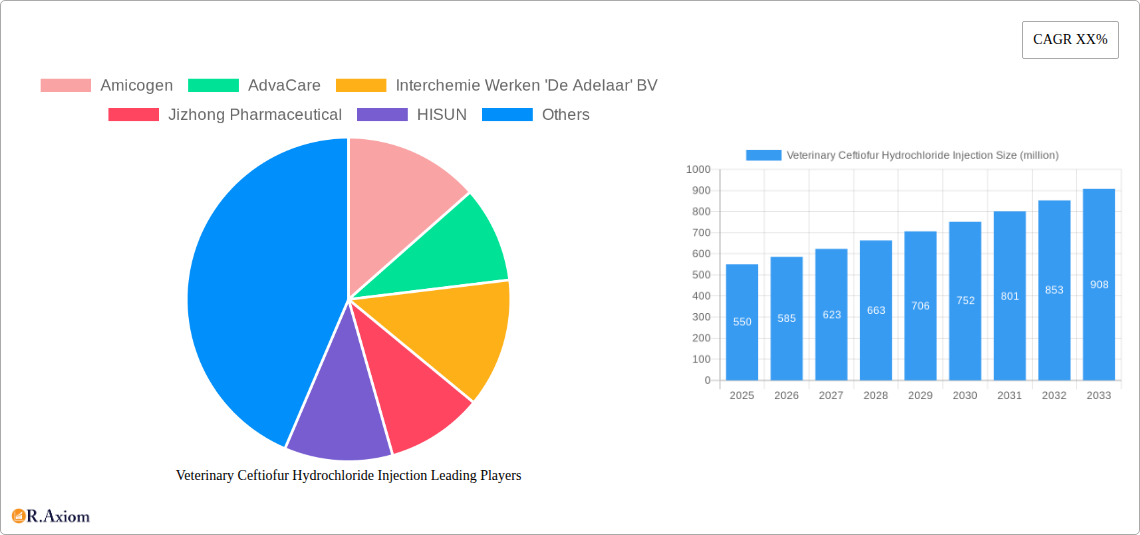
Veterinary Ceftiofur Hydrochloride Injection Market Size (In Million)

Despite these constraints, the market is expected to maintain its growth trajectory due to ongoing research into improving antibiotic stewardship and the increasing awareness among veterinarians about the responsible use of antibiotics. The increasing incidence of bacterial infections resistant to other antibiotics further fuels the demand for ceftiofur hydrochloride. Furthermore, the development of novel formulations and delivery systems is likely to contribute to market expansion. The major players in this market are actively engaged in research and development, expanding their product portfolios, and strategically focusing on emerging markets. This competitive landscape drives innovation and ensures the continued availability of high-quality veterinary ceftiofur hydrochloride injection products.
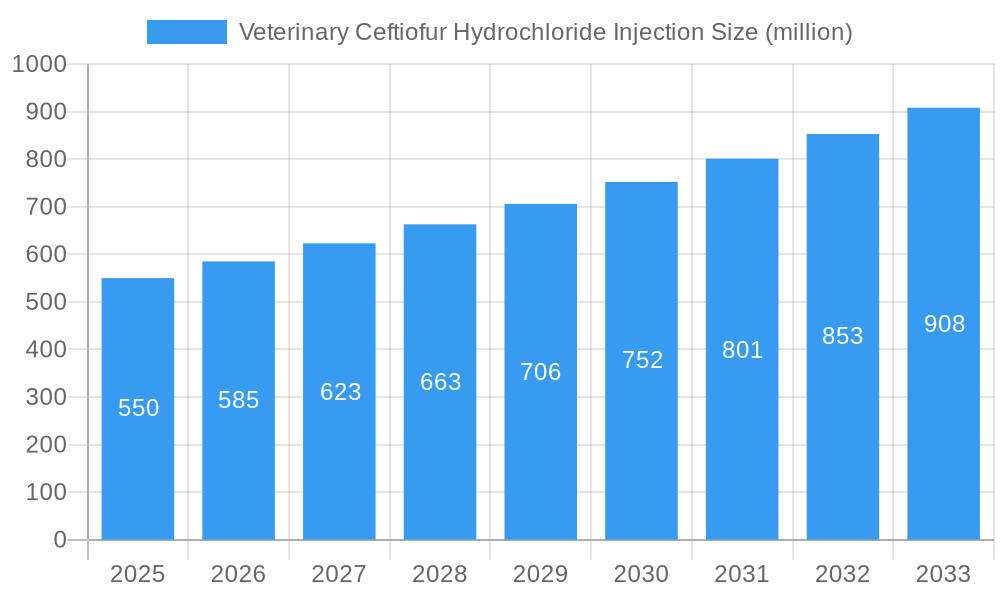
Veterinary Ceftiofur Hydrochloride Injection Company Market Share

Veterinary Ceftiofur Hydrochloride Injection Market Report: 2019-2033
This comprehensive report provides a detailed analysis of the global Veterinary Ceftiofur Hydrochloride Injection market, offering invaluable insights for industry stakeholders, investors, and researchers. Covering the period from 2019 to 2033, with a focus on the 2025-2033 forecast, this report presents a granular view of market dynamics, competitive landscapes, and future growth prospects. The report leverages extensive primary and secondary research to deliver actionable intelligence, allowing businesses to make informed strategic decisions. The global market size in 2025 is estimated at $XX million and is projected to reach $XX million by 2033, exhibiting a CAGR of XX% during the forecast period.
Veterinary Ceftiofur Hydrochloride Injection Market Concentration & Innovation
The global Veterinary Ceftiofur Hydrochloride Injection market exhibits a moderately concentrated structure, with a few major players holding significant market share. However, the presence of numerous smaller companies creates a dynamic competitive landscape. In 2025, the top five companies held approximately XX% of the market share, while the remaining XX% was distributed among numerous smaller players. Innovation is a key driver, with companies focusing on improved formulations, extended release technologies, and enhanced delivery systems. Regulatory frameworks, such as those established by the FDA and EMA, significantly influence market dynamics. Product substitutes, including other antibiotics, also pose a competitive challenge. End-user trends, particularly the increasing demand for effective and safe veterinary treatments, are driving market growth. Over the past five years, the market has witnessed several M&A activities, with deal values exceeding $XX million cumulatively.
- Market Concentration: Top 5 players hold approximately XX% of the market share in 2025.
- Innovation Drivers: Improved formulations, extended release technologies, enhanced delivery systems.
- Regulatory Frameworks: FDA, EMA regulations heavily influence market dynamics.
- Product Substitutes: Other antibiotics pose competitive challenges.
- M&A Activities: Cumulative deal values exceeding $XX million in the last five years.
Veterinary Ceftiofur Hydrochloride Injection Industry Trends & Insights
The Veterinary Ceftiofur Hydrochloride Injection market is experiencing robust growth, fueled by rising animal healthcare expenditure, increasing livestock populations, and a growing awareness of animal welfare. Technological advancements, such as the development of novel formulations and delivery systems, are enhancing the efficacy and convenience of the injection. Consumer preferences are shifting towards more effective and safer products with reduced side effects. Competitive dynamics are intensifying, with companies focusing on product differentiation, brand building, and strategic partnerships. The market experienced significant growth in the historical period (2019-2024), with a CAGR of XX%. This positive trend is expected to continue throughout the forecast period (2025-2033), driven by increasing veterinary practices adopting the injection for various animal diseases. Market penetration in key regions is increasing steadily, with significant untapped potential in emerging economies.
Dominant Markets & Segments in Veterinary Ceftiofur Hydrochloride Injection
The North American market currently dominates the global Veterinary Ceftiofur Hydrochloride Injection market, driven by robust animal healthcare infrastructure, high veterinary spending, and a large livestock population.
- Key Drivers in North America:
- Strong regulatory framework supporting animal health.
- High veterinary spending per capita.
- Large livestock population (cattle, swine, poultry).
- Advanced animal healthcare infrastructure.
The dominance of North America stems from a confluence of factors including high veterinary expenditure, stringent regulatory frameworks that ensure product quality and safety, and a significant livestock population requiring regular veterinary care. Furthermore, the region's advanced animal healthcare infrastructure and strong veterinary practices contribute to higher market penetration. Other regions, like Europe and Asia-Pacific, also exhibit significant growth potential, although at a slower pace compared to North America.
Veterinary Ceftiofur Hydrochloride Injection Product Developments
Recent product innovations in the Veterinary Ceftiofur Hydrochloride Injection market focus on enhancing drug delivery systems, improving bioavailability, and extending the duration of action. These advancements aim to enhance therapeutic efficacy while minimizing side effects, thus increasing market competitiveness. The focus on technological trends allows for better targeted delivery, reducing the overall dosage needed and potentially lowering costs. The market fit of these innovations is reflected in increasing adoption rates within veterinary practices and improved animal health outcomes.
Report Scope & Segmentation Analysis
This report segments the Veterinary Ceftiofur Hydrochloride Injection market by animal type (cattle, swine, poultry, others), dosage form (solution, powder), route of administration (injection), and region (North America, Europe, Asia-Pacific, Rest of the World). Each segment is analyzed in detail, considering growth projections, market size, and competitive dynamics. The projections indicate substantial growth across all segments, with cattle and swine accounting for the largest market share.
Key Drivers of Veterinary Ceftiofur Hydrochloride Injection Growth
The growth of the Veterinary Ceftiofur Hydrochloride Injection market is primarily driven by the increasing prevalence of bacterial infections in livestock, the growing demand for effective and safe antibiotics, and advancements in veterinary technologies. Stringent government regulations regarding animal health and the rising disposable income in developing countries also contribute to this growth. Furthermore, the increasing adoption of preventative healthcare measures in livestock farming and expanding veterinary practices contribute significantly.
Challenges in the Veterinary Ceftiofur Hydrochloride Injection Sector
The Veterinary Ceftiofur Hydrochloride Injection sector faces challenges including the increasing incidence of antibiotic resistance, stringent regulatory approvals, and the potential for adverse effects. Supply chain disruptions and price fluctuations of raw materials also pose significant challenges, potentially impacting production costs and market stability. Intense competition among established and emerging players further complicates market dynamics. These factors can collectively impact market growth and profitability. The estimated impact of these challenges on the market in 2025 is a reduced growth rate of XX%.
Emerging Opportunities in Veterinary Ceftiofur Hydrochloride Injection
Emerging opportunities lie in the development of novel formulations to combat antibiotic resistance, exploring new drug delivery systems, and expanding into untapped markets in developing economies. The increasing demand for customized veterinary solutions and the growing use of precision medicine in animal healthcare offer promising avenues for growth. Focusing on sustainable and environmentally friendly manufacturing processes can also enhance market competitiveness.
Leading Players in the Veterinary Ceftiofur Hydrochloride Injection Market
- Amicogen
- AdvaCare
- Interchemie Werken 'De Adelaar' BV
- Jizhong Pharmaceutical
- HISUN
- HVSEN BIOTECH
- North China Pharmaceutical Group
- Chengdu Xinheng Pharmaceutical
- SMAPC
- Henan Guozhu Biotechnology
- HENAN MU JIN BIOLOGICAL TECHNOLOGY
Key Developments in Veterinary Ceftiofur Hydrochloride Injection Industry
- 2022 Q4: AdvaCare announced the launch of a new, improved formulation of Veterinary Ceftiofur Hydrochloride Injection.
- 2023 Q1: Amicogen and HISUN formed a strategic partnership to expand distribution networks.
- 2024 Q2: Jizhong Pharmaceutical received regulatory approval for a novel extended-release formulation. (Further developments with specific dates and impacts need to be added based on actual market data.)
Strategic Outlook for Veterinary Ceftiofur Hydrochloride Injection Market
The Veterinary Ceftiofur Hydrochloride Injection market is poised for significant growth in the coming years, driven by increasing demand, technological advancements, and expanding veterinary services. Opportunities exist in developing innovative products, expanding into new markets, and strengthening partnerships to enhance market penetration and achieve sustainable growth. Companies focusing on product differentiation, cost-effectiveness, and regulatory compliance will gain a competitive edge in this evolving market.
Veterinary Ceftiofur Hydrochloride Injection Segmentation
-
1. Application
- 1.1. Pig
- 1.2. Cattle
- 1.3. Sheep
- 1.4. Others
-
2. Types
- 2.1. Crystal Injection
- 2.2. Oily Suspension
Veterinary Ceftiofur Hydrochloride Injection Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
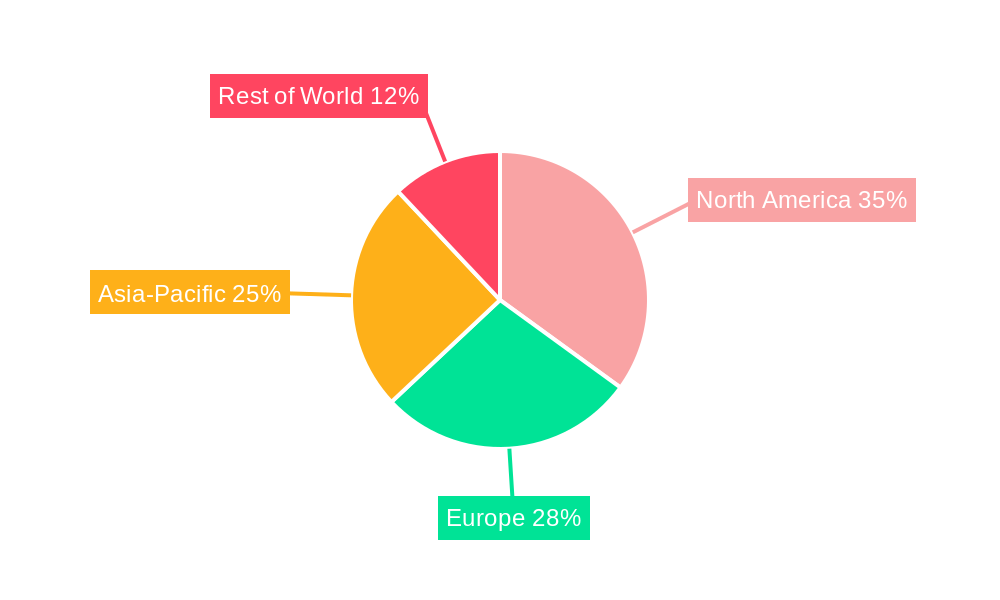
Veterinary Ceftiofur Hydrochloride Injection Regional Market Share

Geographic Coverage of Veterinary Ceftiofur Hydrochloride Injection
Veterinary Ceftiofur Hydrochloride Injection REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.5% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. RAX Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Pig
- 5.1.2. Cattle
- 5.1.3. Sheep
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Crystal Injection
- 5.2.2. Oily Suspension
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Veterinary Ceftiofur Hydrochloride Injection Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Pig
- 6.1.2. Cattle
- 6.1.3. Sheep
- 6.1.4. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Crystal Injection
- 6.2.2. Oily Suspension
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Veterinary Ceftiofur Hydrochloride Injection Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Pig
- 7.1.2. Cattle
- 7.1.3. Sheep
- 7.1.4. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Crystal Injection
- 7.2.2. Oily Suspension
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Veterinary Ceftiofur Hydrochloride Injection Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Pig
- 8.1.2. Cattle
- 8.1.3. Sheep
- 8.1.4. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Crystal Injection
- 8.2.2. Oily Suspension
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Veterinary Ceftiofur Hydrochloride Injection Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Pig
- 9.1.2. Cattle
- 9.1.3. Sheep
- 9.1.4. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Crystal Injection
- 9.2.2. Oily Suspension
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Veterinary Ceftiofur Hydrochloride Injection Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Pig
- 10.1.2. Cattle
- 10.1.3. Sheep
- 10.1.4. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Crystal Injection
- 10.2.2. Oily Suspension
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Veterinary Ceftiofur Hydrochloride Injection Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Pig
- 11.1.2. Cattle
- 11.1.3. Sheep
- 11.1.4. Others
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Crystal Injection
- 11.2.2. Oily Suspension
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Amicogen
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 AdvaCare
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Interchemie Werken 'De Adelaar' BV
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Jizhong Pharmaceutical
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 HISUN
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 HVSEN BIOTECH
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 North China Pharmaceutical Group
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Chengdu Xinheng Pharmaceutical
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 SMAPC
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Henan Guozhu Biotechnology
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 HENAN MU JIN BIOLOGICAL TECHNOLOGY
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.1 Amicogen
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Veterinary Ceftiofur Hydrochloride Injection Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Application 2025 & 2033
- Figure 3: North America Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Types 2025 & 2033
- Figure 5: North America Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Country 2025 & 2033
- Figure 7: North America Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Application 2025 & 2033
- Figure 9: South America Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Types 2025 & 2033
- Figure 11: South America Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Country 2025 & 2033
- Figure 13: South America Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Application 2025 & 2033
- Figure 15: Europe Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Types 2025 & 2033
- Figure 17: Europe Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Country 2025 & 2033
- Figure 19: Europe Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Application 2025 & 2033
- Figure 21: Middle East & Africa Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Types 2025 & 2033
- Figure 23: Middle East & Africa Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Country 2025 & 2033
- Figure 25: Middle East & Africa Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Application 2025 & 2033
- Figure 27: Asia Pacific Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Types 2025 & 2033
- Figure 29: Asia Pacific Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Veterinary Ceftiofur Hydrochloride Injection Revenue (million), by Country 2025 & 2033
- Figure 31: Asia Pacific Veterinary Ceftiofur Hydrochloride Injection Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Types 2020 & 2033
- Table 3: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Region 2020 & 2033
- Table 4: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Application 2020 & 2033
- Table 5: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Types 2020 & 2033
- Table 6: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Country 2020 & 2033
- Table 7: United States Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 8: Canada Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 9: Mexico Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Application 2020 & 2033
- Table 11: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Types 2020 & 2033
- Table 12: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Country 2020 & 2033
- Table 13: Brazil Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: Argentina Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Application 2020 & 2033
- Table 17: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Types 2020 & 2033
- Table 18: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Germany Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 21: France Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Italy Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 23: Spain Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 24: Russia Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 25: Benelux Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: Nordics Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Application 2020 & 2033
- Table 29: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Types 2020 & 2033
- Table 30: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Country 2020 & 2033
- Table 31: Turkey Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Israel Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 33: GCC Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: North Africa Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 35: South Africa Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 37: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Application 2020 & 2033
- Table 38: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Types 2020 & 2033
- Table 39: Global Veterinary Ceftiofur Hydrochloride Injection Revenue million Forecast, by Country 2020 & 2033
- Table 40: China Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 41: India Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Japan Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 43: South Korea Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 45: Oceania Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Veterinary Ceftiofur Hydrochloride Injection Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Veterinary Ceftiofur Hydrochloride Injection?
The projected CAGR is approximately 5.5%.
2. Which companies are prominent players in the Veterinary Ceftiofur Hydrochloride Injection?
Key companies in the market include Amicogen, AdvaCare, Interchemie Werken 'De Adelaar' BV, Jizhong Pharmaceutical, HISUN, HVSEN BIOTECH, North China Pharmaceutical Group, Chengdu Xinheng Pharmaceutical, SMAPC, Henan Guozhu Biotechnology, HENAN MU JIN BIOLOGICAL TECHNOLOGY.
3. What are the main segments of the Veterinary Ceftiofur Hydrochloride Injection?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 500 million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Veterinary Ceftiofur Hydrochloride Injection," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Veterinary Ceftiofur Hydrochloride Injection report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Veterinary Ceftiofur Hydrochloride Injection?
To stay informed about further developments, trends, and reports in the Veterinary Ceftiofur Hydrochloride Injection, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
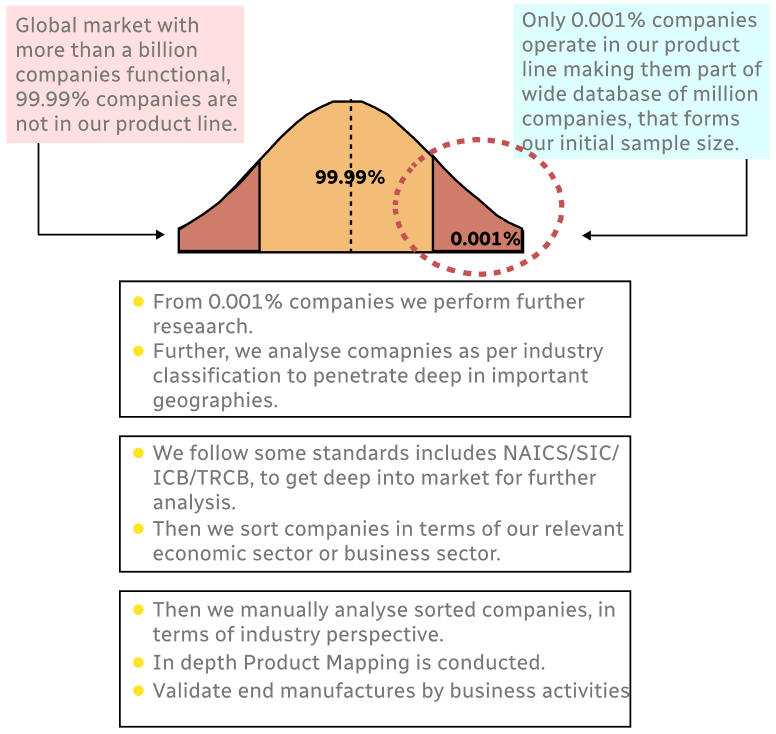
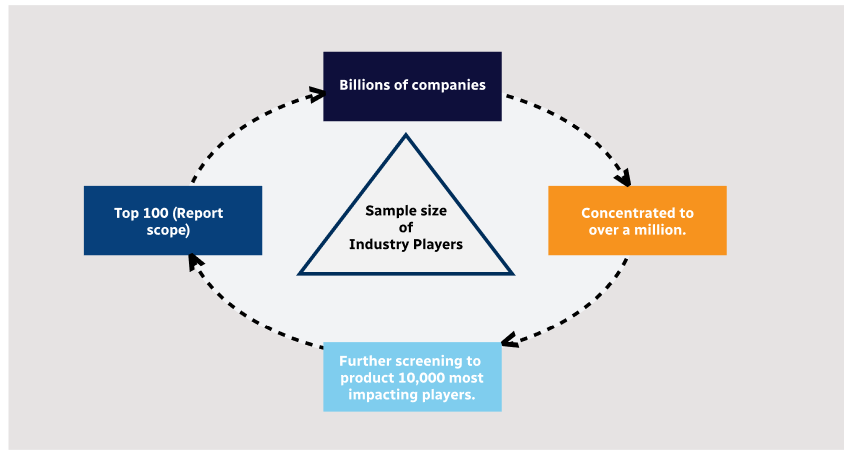
Step 1 - Identification of Relevant Samples Size from Population Database
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
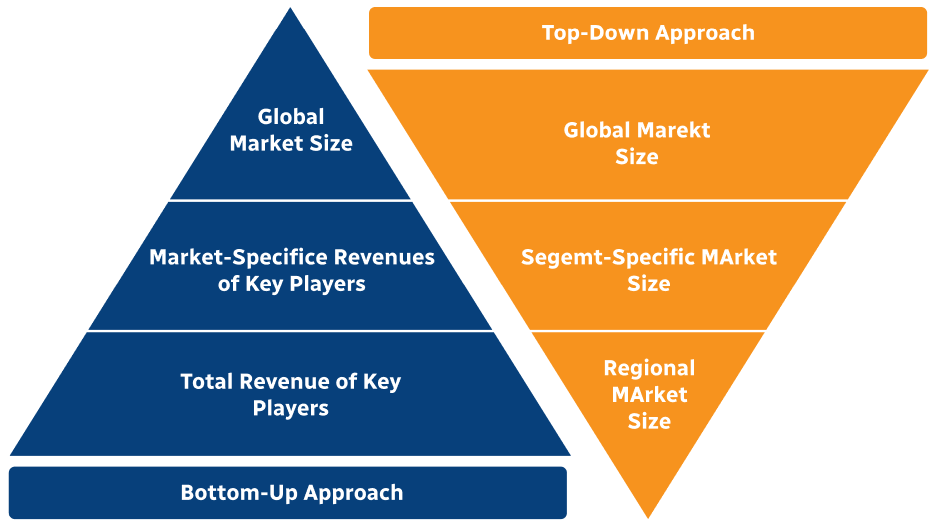
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence