Key Insights
The medical device analytical testing outsourcing market is experiencing robust expansion, projected to reach $8.6 billion in 2024, with an impressive Compound Annual Growth Rate (CAGR) of 8.8% during the forecast period of 2025-2033. This growth is fueled by several critical drivers. The increasing complexity of medical devices, coupled with stringent regulatory requirements from bodies like the FDA and EMA, necessitates specialized analytical testing to ensure safety, efficacy, and compliance. Furthermore, the escalating cost of in-house laboratory infrastructure and the need for specialized expertise are compelling medical device manufacturers, both small and large enterprises, to outsource these critical functions. The trend towards miniaturization and the development of novel materials for medical devices also demand advanced characterization and testing capabilities that are often more efficiently accessed through outsourcing partners. The expanding healthcare sector globally and the continuous innovation in medical technology further contribute to the sustained demand for these services.
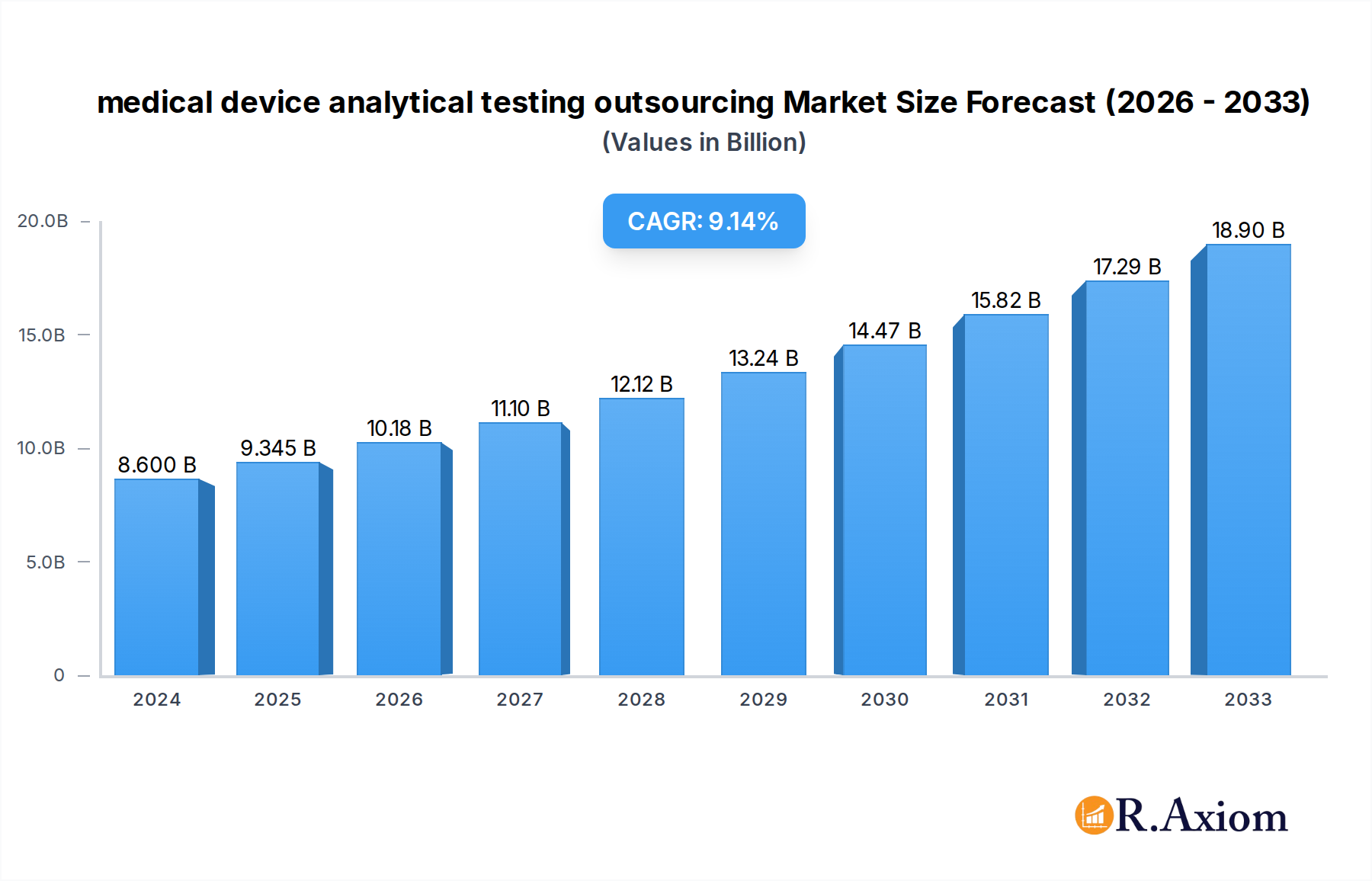
medical device analytical testing outsourcing Market Size (In Billion)

The market is segmented across various crucial testing types, including extractable and leachable studies, material characterization, physical testing, bioburden testing, and sterility testing. These specialized services are vital for ensuring the safety and performance of a wide range of medical devices. Emerging trends like the increasing adoption of advanced analytical techniques, such as mass spectrometry and chromatography, along with the growing focus on biocompatibility and toxicology assessments, are shaping the outsourcing landscape. While the market demonstrates strong growth, potential restraints include the high cost of advanced analytical equipment and the need for highly skilled personnel, which can influence outsourcing decisions for some companies. The competitive landscape features prominent global players like SGS SA, Eurofins Scientific, and Intertek Group, all vying to provide comprehensive solutions to meet the evolving needs of the medical device industry across key regions.
medical device analytical testing outsourcing Company Market Share

medical device analytical testing outsourcing Market Concentration & Innovation
The medical device analytical testing outsourcing market exhibits moderate concentration, with a few dominant players alongside numerous specialized service providers. Key companies such as SGS SA (Switzerland), Toxikon, Inc. (US), Eurofins Scientific (Luxembourg), Pace Analytical Services, LLC (US), Intertek Group plc (UK), WuXi AppTec (China), NORTH AMERICAN SCIENCE ASSOCIATES INC. (US), Envigo (UK), Charles River Laboratories International Inc. (US), and Medical Device Testing Services (US) collectively hold a significant market share, estimated at over 80 billion. Innovation is primarily driven by increasingly stringent regulatory requirements from bodies like the FDA and EMA, pushing CROs to invest in advanced analytical techniques and capabilities for extractable and leachable testing, material characterization, and bioburden testing. The constant evolution of medical device materials and designs necessitates continuous development in physical testing methods and sterility testing protocols. Regulatory frameworks are a critical influence, dictating the types and rigor of testing required for device approval, thereby shaping service offerings. Product substitutes are minimal, as specialized analytical testing cannot be easily replicated in-house by many medical device manufacturers, particularly small and medium enterprises (SMEs). End-user trends indicate a growing demand for comprehensive testing solutions, from early-stage R&D to post-market surveillance, with a preference for integrated service providers. Mergers and acquisitions (M&A) activities are prevalent, with deal values in the multi-billion range, as larger players aim to expand their service portfolios, geographical reach, and client base. For instance, acquisitions of smaller, specialized labs by major CROs are common to gain access to niche expertise or customer segments.
medical device analytical testing outsourcing Industry Trends & Insights
The medical device analytical testing outsourcing market is experiencing robust growth, fueled by a confluence of factors that are reshaping how medical device companies approach product development and compliance. The projected Compound Annual Growth Rate (CAGR) for this sector is estimated to be around 8.5 billion, indicating a significant expansion over the forecast period of 2025–2033. Market penetration is rising as more medical device manufacturers, particularly small and medium enterprises (SMEs), recognize the strategic advantages of outsourcing. This trend is driven by the escalating complexity of regulatory requirements, the need for specialized expertise, and the significant capital investment required for in-house testing laboratories. Technological disruptions are playing a pivotal role, with advancements in areas such as mass spectrometry, chromatography, and advanced imaging techniques enhancing the precision and efficiency of extractable and leachable testing and material characterization. The ability to detect and quantify trace amounts of leachable substances is becoming paramount, especially for implantable devices and drug-eluting technologies. Consumer preferences are indirectly influencing the market by demanding safer and more effective medical devices, which in turn necessitates more comprehensive and rigorous analytical testing. This translates to an increased demand for validated testing methods and robust data integrity. Competitive dynamics are intensifying, with established players like SGS SA, Eurofins Scientific, and WuXi AppTec investing heavily in expanding their capabilities and global footprints. Emerging players are also carving out niches by focusing on specialized testing services or catering to specific device categories. The growing outsourcing trend allows medical device companies to accelerate their time-to-market, reduce operational costs, and focus on core competencies, all while ensuring compliance with evolving global regulatory standards. The increasing outsourcing of physical testing and bioburden testing further underscores the market's growth trajectory.
Dominant Markets & Segments in medical device analytical testing outsourcing
The medical device analytical testing outsourcing market is characterized by distinct dominant regions and segments that are driving its overall expansion. North America, particularly the United States, is a leading region due to the high concentration of medical device manufacturers, robust regulatory infrastructure, and significant R&D investments. The presence of major industry players like Toxikon, Inc., Pace Analytical Services, LLC, Charles River Laboratories International Inc., and NORTH AMERICAN SCIENCE ASSOCIATES INC. further solidifies its dominance. Economic policies that encourage innovation and stringent regulatory oversight from the FDA create a fertile ground for analytical testing services.
Within the Application segmentation, Large Enterprise clients represent a dominant segment. These companies possess a higher volume of medical devices, a greater need for comprehensive testing across the product lifecycle, and the financial capacity to invest in specialized outsourcing. Their extensive product portfolios and global market presence necessitate extensive validation and compliance testing, including rigorous extractable and leachable testing and material characterization.
In terms of Types, Extractable & Leachable Testing stands out as a dominant segment. This is driven by increasingly stringent regulatory demands, particularly concerning patient safety and the potential for adverse reactions from materials used in medical devices. Devices intended for prolonged patient contact, implantable devices, and drug-delivery systems are subject to intense scrutiny for leachables, making this a critical area of outsourcing. Companies like SGS SA and Eurofins Scientific are heavily invested in providing advanced E&L solutions.
Material Characterization is another highly dominant segment. Understanding the chemical composition, physical properties, and potential degradation pathways of materials used in medical devices is crucial for ensuring device performance, safety, and longevity. This segment encompasses a wide range of analytical techniques and is essential for every stage of device development.
Physical Testing also holds significant importance, covering aspects like mechanical strength, durability, biocompatibility, and electrical safety. This is a non-negotiable aspect of medical device development, and outsourcing allows manufacturers to access specialized equipment and expertise.
Bioburden Testing and Sterility Testing are critical for ensuring the safety of devices, especially those that are invasive or come into contact with sterile bodily tissues. The regulatory emphasis on preventing infections has amplified the demand for these testing services.
The Others segment, while broad, can include specialized testing like elemental impurity analysis, particulate matter testing, and advanced microscopy, all of which contribute to the overall safety and efficacy of medical devices and are often outsourced due to their highly specialized nature.
medical device analytical testing outsourcing Product Developments
Product developments in medical device analytical testing outsourcing are characterized by an increasing focus on advanced analytical techniques, automation, and integrated service offerings. Companies are investing in cutting-edge technologies for extractable and leachable testing, such as high-resolution mass spectrometry (HRMS), to achieve lower detection limits and more accurate identification of potential contaminants. Innovations in material characterization include the application of advanced spectroscopic methods and microscopy to gain deeper insights into material composition and performance. Furthermore, the development of automated platforms for bioburden testing and sterility testing is enhancing throughput and reducing turnaround times. These advancements offer competitive advantages by enabling faster product development cycles, ensuring compliance with evolving regulations, and ultimately leading to safer and more reliable medical devices for patients.
Report Scope & Segmentation Analysis
This report delves into the medical device analytical testing outsourcing market, providing a comprehensive analysis across several key segmentation dimensions. The Application segmentation includes Small & Medium Enterprise and Large Enterprise. The Small & Medium Enterprise segment, while smaller in individual contract value, collectively represents a significant and growing market due to the increasing trend of outsourcing by these entities seeking cost-effective and expert solutions. The Large Enterprise segment dominates in terms of overall market value due to the sheer volume and complexity of testing required for their extensive product portfolios.
The Types segmentation encompasses Extractable & Leachable, Material Characterization, Physical Testing, Bioburden Testing, Sterility Testing, and Others. Extractable & Leachable testing is projected for substantial growth owing to stringent regulatory scrutiny and patient safety concerns. Material Characterization is a foundational segment with consistent demand for understanding device materials. Physical Testing remains a critical component, with ongoing innovations in methodologies. Bioburden and Sterility Testing are indispensable for safety compliance, especially for invasive devices, and are expected to see steady growth. The Others category, encompassing specialized analyses, is anticipated to grow as device complexity increases.
Key Drivers of medical device analytical testing outsourcing Growth
The medical device analytical testing outsourcing market is propelled by several key drivers. Firstly, the increasingly complex and evolving global regulatory landscape, mandated by authorities such as the FDA and EMA, necessitates specialized expertise and rigorous testing that many manufacturers find more efficient to outsource. Secondly, the continuous innovation in medical device technology, leading to novel materials and complex designs, requires advanced analytical capabilities that are often beyond the scope of in-house laboratories. Thirdly, the drive for cost optimization and faster time-to-market encourages companies, particularly SMEs, to leverage the economies of scale and specialized infrastructure offered by CROs. Lastly, the growing emphasis on patient safety and product quality worldwide amplifies the demand for comprehensive testing, including extractable and leachable testing, material characterization, and sterility testing.
Challenges in the medical device analytical testing outsourcing Sector
Despite the robust growth, the medical device analytical testing outsourcing sector faces several challenges. A primary restraint is the stringent and ever-changing regulatory environment, which requires continuous investment in updating methodologies and compliance protocols. Another significant hurdle is the potential for supply chain disruptions, which can impact the availability of specialized reagents and equipment, thereby affecting turnaround times. Fierce competition among service providers can lead to price pressures, impacting profit margins, especially for smaller CROs. Furthermore, ensuring data integrity and intellectual property protection for clients is a paramount concern that requires robust cybersecurity measures and strict operational protocols. Miscommunication or a lack of clear understanding of project scope between clients and service providers can also lead to delays and increased costs.
Emerging Opportunities in medical device analytical testing outsourcing
Emerging opportunities in the medical device analytical testing outsourcing market are abundant, driven by technological advancements and shifting industry demands. The growing field of personalized medicine and the development of advanced biologics and combination devices present new avenues for specialized analytical testing. Increased adoption of artificial intelligence (AI) and machine learning (ML) in data analysis can offer more predictive insights and faster identification of potential issues. Expansion into emerging geographical markets with growing medical device manufacturing bases also presents significant growth potential. Furthermore, the increasing demand for sustainability in device manufacturing is creating opportunities for testing related to the environmental impact and lifecycle assessment of medical devices. The integration of extractable and leachable testing with toxicological assessments is another area of increasing demand.
Leading Players in the medical device analytical testing outsourcing Market
- SGS SA
- Toxikon, Inc.
- Eurofins Scientific
- Pace Analytical Services, LLC
- Intertek Group plc
- WuXi AppTec
- NORTH AMERICAN SCIENCE ASSOCIATES INC.
- Envigo
- Charles River Laboratories International Inc.
- Medical Device Testing Services
Key Developments in medical device analytical testing outsourcing Industry
- 2023/09: SGS SA expands its advanced analytical capabilities in Europe to support novel medical device materials.
- 2023/07: Toxikon, Inc. announces enhanced capacity for extractable and leachable studies, meeting increased demand.
- 2023/05: Eurofins Scientific acquires a specialized medical device testing laboratory in Asia, broadening its global reach.
- 2023/03: Pace Analytical Services, LLC invests in new high-resolution mass spectrometry equipment for comprehensive material characterization.
- 2023/01: Intertek Group plc launches a new suite of services for biocompatibility testing of implantable medical devices.
- 2022/11: WuXi AppTec strengthens its bioburden and sterility testing services to cater to sterile medical device manufacturers.
- 2022/09: NORTH AMERICAN SCIENCE ASSOCIATES INC. partners with a leading medical device manufacturer to streamline product validation testing.
- 2022/07: Envigo enhances its physical testing capabilities for a wide range of medical device components.
- 2022/05: Charles River Laboratories International Inc. announces expansion of its preclinical testing services for drug-eluting medical devices.
- 2022/03: Medical Device Testing Services introduces a comprehensive testing package for wearable medical technology.
Strategic Outlook for medical device analytical testing outsourcing Market
The strategic outlook for the medical device analytical testing outsourcing market remains exceptionally positive. Growth catalysts include the sustained pressure from global regulatory bodies for enhanced patient safety and product efficacy, driving continuous demand for sophisticated extractable and leachable testing, material characterization, and sterility testing. The accelerating pace of medical device innovation, particularly in areas like minimally invasive surgery, digital health, and advanced biomaterials, will necessitate specialized analytical support. Companies that can offer integrated, end-to-end testing solutions, from early-stage R&D to post-market surveillance, will gain a significant competitive advantage. Strategic investments in advanced technologies, geographic expansion into high-growth regions, and a focus on niche service offerings will be crucial for market participants to capitalize on the expanding opportunities and maintain a strong market position in the coming years.
medical device analytical testing outsourcing Segmentation
-
1. Application
- 1.1. Small & Medium Enterprise
- 1.2. Large Enterprise
-
2. Types
- 2.1. Extractable & Leachable
- 2.2. Material Charecterization
- 2.3. Physical Testing
- 2.4. Bioburden Testing
- 2.5. Sterility Testing
- 2.6. Others
medical device analytical testing outsourcing Segmentation By Geography
- 1. CA
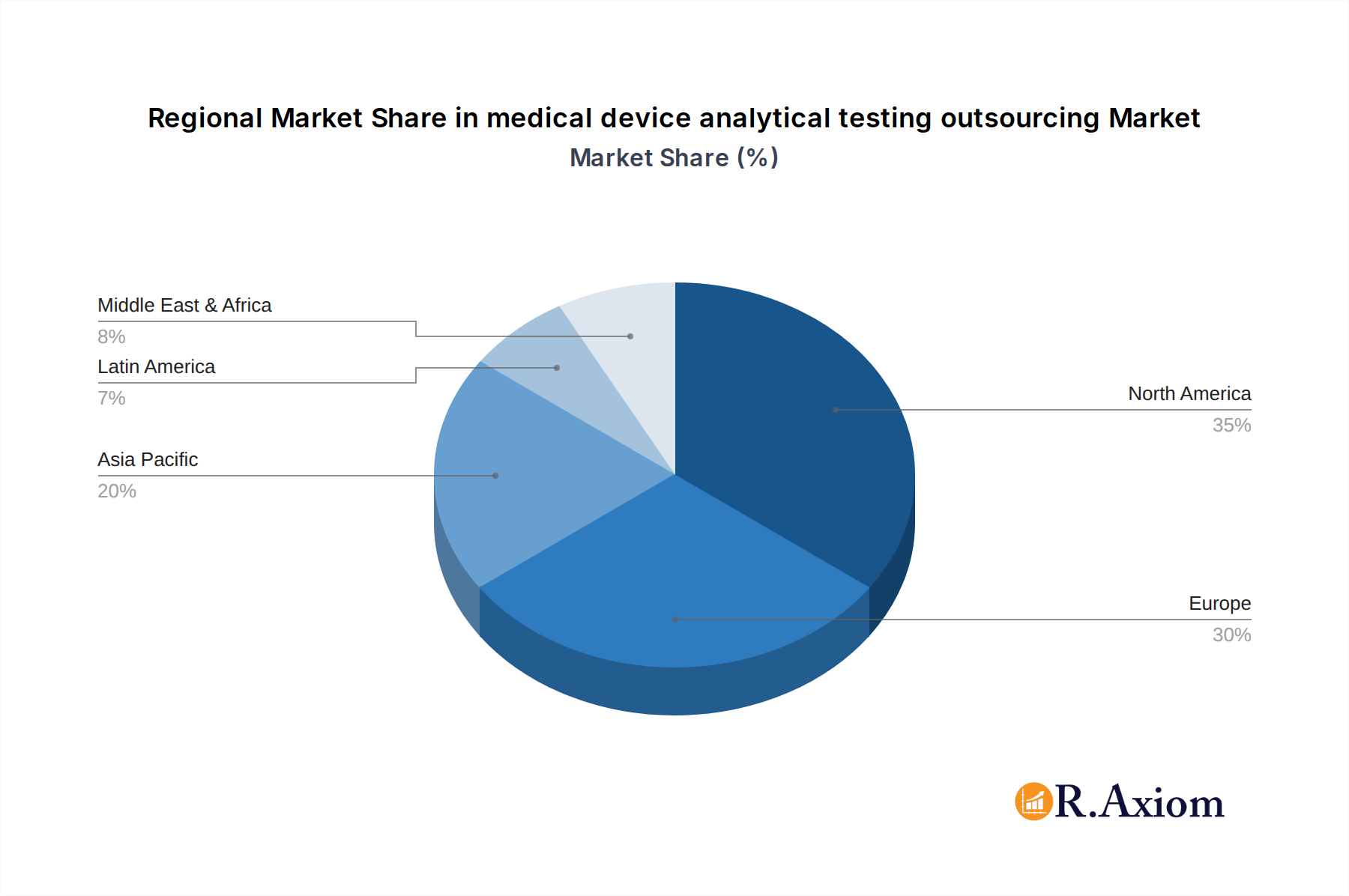
medical device analytical testing outsourcing Regional Market Share

Geographic Coverage of medical device analytical testing outsourcing
medical device analytical testing outsourcing REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.8% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. medical device analytical testing outsourcing Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Small & Medium Enterprise
- 5.1.2. Large Enterprise
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Extractable & Leachable
- 5.2.2. Material Charecterization
- 5.2.3. Physical Testing
- 5.2.4. Bioburden Testing
- 5.2.5. Sterility Testing
- 5.2.6. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. CA
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Competitive Analysis
- 6.1. Market Share Analysis 2025
- 6.2. Company Profiles
- 6.2.1 SGS SA (Switzerland)
- 6.2.1.1. Overview
- 6.2.1.2. Products
- 6.2.1.3. SWOT Analysis
- 6.2.1.4. Recent Developments
- 6.2.1.5. Financials (Based on Availability)
- 6.2.2 Toxikon
- 6.2.2.1. Overview
- 6.2.2.2. Products
- 6.2.2.3. SWOT Analysis
- 6.2.2.4. Recent Developments
- 6.2.2.5. Financials (Based on Availability)
- 6.2.3 Inc. (US)
- 6.2.3.1. Overview
- 6.2.3.2. Products
- 6.2.3.3. SWOT Analysis
- 6.2.3.4. Recent Developments
- 6.2.3.5. Financials (Based on Availability)
- 6.2.4 Eurofins Scientific (Luxembourg)
- 6.2.4.1. Overview
- 6.2.4.2. Products
- 6.2.4.3. SWOT Analysis
- 6.2.4.4. Recent Developments
- 6.2.4.5. Financials (Based on Availability)
- 6.2.5 Pace Analytical Services
- 6.2.5.1. Overview
- 6.2.5.2. Products
- 6.2.5.3. SWOT Analysis
- 6.2.5.4. Recent Developments
- 6.2.5.5. Financials (Based on Availability)
- 6.2.6 LLC (US)
- 6.2.6.1. Overview
- 6.2.6.2. Products
- 6.2.6.3. SWOT Analysis
- 6.2.6.4. Recent Developments
- 6.2.6.5. Financials (Based on Availability)
- 6.2.7 Intertek Group plc (UK)
- 6.2.7.1. Overview
- 6.2.7.2. Products
- 6.2.7.3. SWOT Analysis
- 6.2.7.4. Recent Developments
- 6.2.7.5. Financials (Based on Availability)
- 6.2.8 WuXi AppTec. (China)
- 6.2.8.1. Overview
- 6.2.8.2. Products
- 6.2.8.3. SWOT Analysis
- 6.2.8.4. Recent Developments
- 6.2.8.5. Financials (Based on Availability)
- 6.2.9 NORTH AMERICAN SCIENCE ASSOCIATES INC. (US)
- 6.2.9.1. Overview
- 6.2.9.2. Products
- 6.2.9.3. SWOT Analysis
- 6.2.9.4. Recent Developments
- 6.2.9.5. Financials (Based on Availability)
- 6.2.10 Envigo (UK)
- 6.2.10.1. Overview
- 6.2.10.2. Products
- 6.2.10.3. SWOT Analysis
- 6.2.10.4. Recent Developments
- 6.2.10.5. Financials (Based on Availability)
- 6.2.11 Charles River Laboratories International Inc. (US)
- 6.2.11.1. Overview
- 6.2.11.2. Products
- 6.2.11.3. SWOT Analysis
- 6.2.11.4. Recent Developments
- 6.2.11.5. Financials (Based on Availability)
- 6.2.12 Medical Device Testing Services (US)
- 6.2.12.1. Overview
- 6.2.12.2. Products
- 6.2.12.3. SWOT Analysis
- 6.2.12.4. Recent Developments
- 6.2.12.5. Financials (Based on Availability)
- 6.2.1 SGS SA (Switzerland)
List of Figures
- Figure 1: medical device analytical testing outsourcing Revenue Breakdown (undefined, %) by Product 2025 & 2033
- Figure 2: medical device analytical testing outsourcing Share (%) by Company 2025
List of Tables
- Table 1: medical device analytical testing outsourcing Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: medical device analytical testing outsourcing Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: medical device analytical testing outsourcing Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: medical device analytical testing outsourcing Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: medical device analytical testing outsourcing Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: medical device analytical testing outsourcing Revenue undefined Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the medical device analytical testing outsourcing?
The projected CAGR is approximately 8.8%.
2. Which companies are prominent players in the medical device analytical testing outsourcing?
Key companies in the market include SGS SA (Switzerland), Toxikon, Inc. (US), Eurofins Scientific (Luxembourg), Pace Analytical Services, LLC (US), Intertek Group plc (UK), WuXi AppTec. (China), NORTH AMERICAN SCIENCE ASSOCIATES INC. (US), Envigo (UK), Charles River Laboratories International Inc. (US), Medical Device Testing Services (US).
3. What are the main segments of the medical device analytical testing outsourcing?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3400.00, USD 5100.00, and USD 6800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "medical device analytical testing outsourcing," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the medical device analytical testing outsourcing report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the medical device analytical testing outsourcing?
To stay informed about further developments, trends, and reports in the medical device analytical testing outsourcing, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
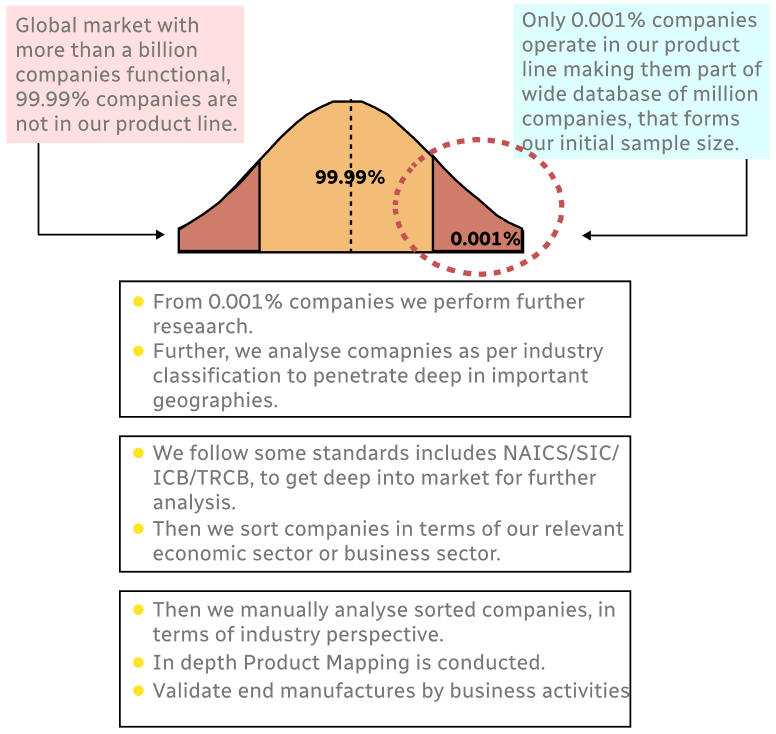
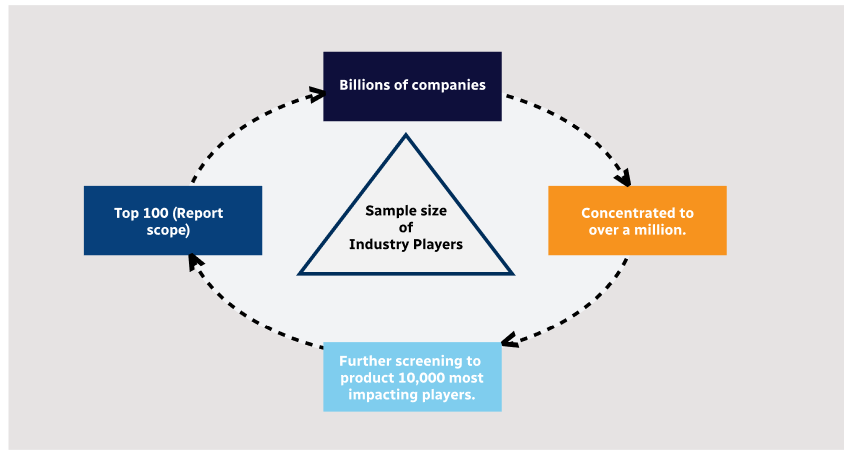
Step 1 - Identification of Relevant Samples Size from Population Database
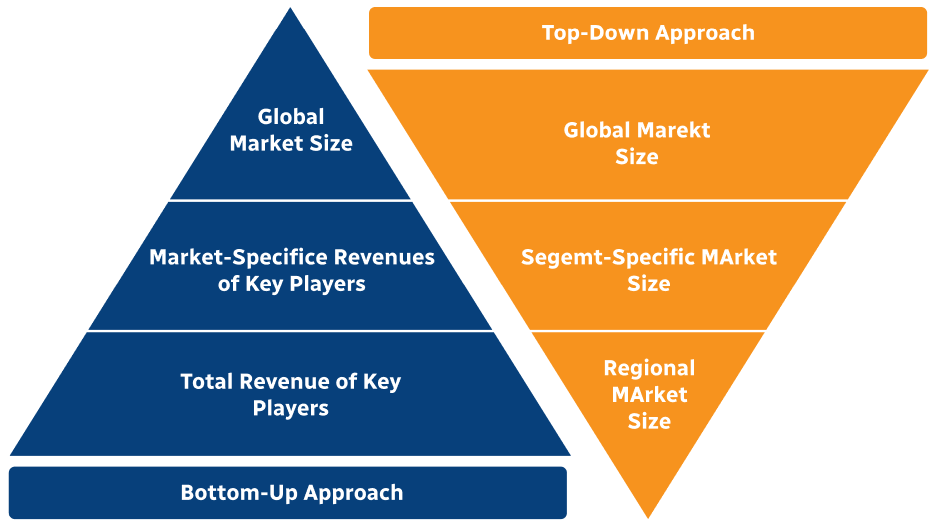
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence