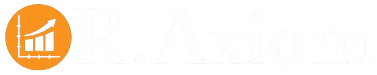Key Insights
The global market for Polymers for Implantable Medical Devices is poised for robust growth, currently valued at an estimated \$1447 million. This expansion is driven by a confluence of factors, including an aging global population that demands more sophisticated medical interventions, a rising incidence of chronic diseases requiring long-term implantable solutions, and continuous advancements in polymer science leading to the development of biocompatible, durable, and functional materials. Key applications benefiting from these advancements include orthopedics, cardiovascular devices, and neurostimulation, where polymers offer superior performance and patient outcomes compared to traditional materials. The market's projected Compound Annual Growth Rate (CAGR) of 3.8% underscores its steady and significant expansion trajectory, fueled by increased healthcare spending and the growing preference for minimally invasive procedures that often necessitate advanced polymer-based implants.
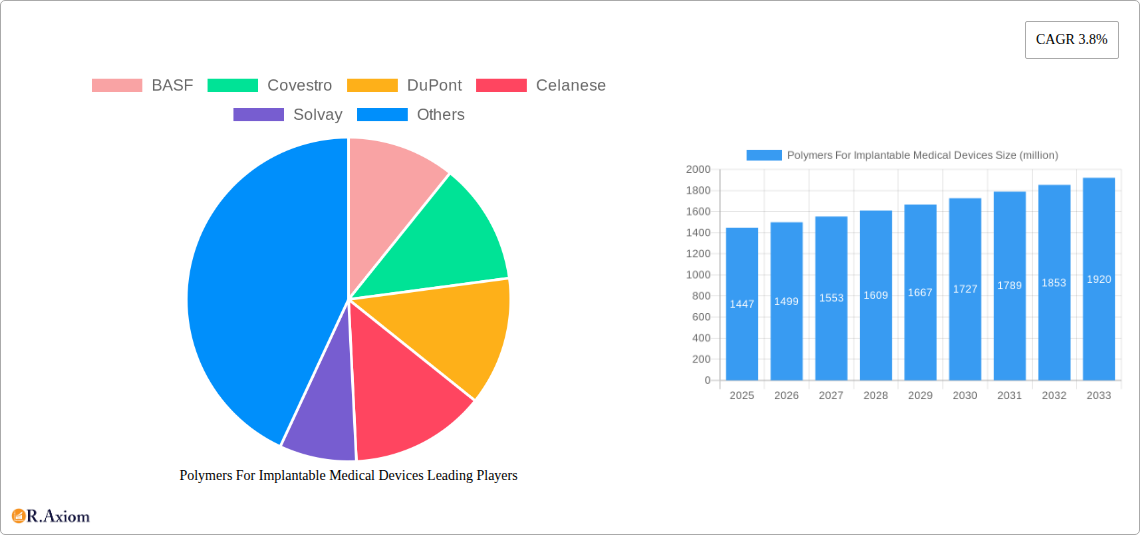
Polymers For Implantable Medical Devices Market Size (In Billion)

The market is segmented into various polymer types, with Polymeric materials dominating due to their versatility, cost-effectiveness, and tunable properties. Within applications, Acetal (POM), Acrylics (including hydrogels and MMA/PMMA), and Fluorocarbons are witnessing substantial demand, each offering unique advantages for specific medical device requirements. For instance, Acetal excels in load-bearing orthopedic implants, while Fluorocarbons are favored for their chemical inertness in cardiovascular applications. The forecast period from 2025 to 2033 is expected to witness further innovation, with a growing emphasis on bioresorbable polymers and smart materials that can actively interact with the body, further expanding the application landscape and market value. Leading companies such as BASF, Covestro, and DuPont are at the forefront of this innovation, investing heavily in research and development to introduce next-generation implantable polymer solutions and capture a significant share of this dynamic and evolving market.
Polymers For Implantable Medical Devices Company Market Share

Polymers For Implantable Medical Devices Market Concentration & Innovation
The global polymers for implantable medical devices market exhibits a dynamic landscape characterized by moderate to high concentration among key players and continuous innovation driven by stringent regulatory demands and advancements in biomaterial science. Major companies such as BASF, Covestro, DuPont, Celanese, and Solvay are at the forefront, investing heavily in research and development to create novel polymeric materials with enhanced biocompatibility, biodegradability, and mechanical properties. This pursuit of innovation is crucial for addressing the evolving needs of various medical applications, from orthopedic implants to cardiovascular devices. Regulatory frameworks, including FDA and EMA approvals, significantly influence market entry and product development, necessitating extensive testing and validation. The threat of product substitutes, while present, is mitigated by the specialized nature and performance requirements of implantable devices, where advanced polymers often outperform traditional materials. End-user trends favor minimally invasive procedures and personalized medicine, pushing for the development of smaller, more advanced, and potentially bioresorbable polymer-based implants. Merger and acquisition (M&A) activities are strategic maneuvers employed by leading players to consolidate market share, acquire innovative technologies, and expand their product portfolios. For instance, M&A deals in the past three years have ranged from xx million to xx million, indicating a proactive approach to strategic growth. The market share of top-tier companies collectively stands at approximately 60-70%, underscoring the concentrated nature of this specialized industry.
Polymers For Implantable Medical Devices Industry Trends & Insights
The polymers for implantable medical devices market is experiencing robust growth, fueled by an aging global population, a rising prevalence of chronic diseases, and continuous technological advancements in minimally invasive surgical techniques. The projected Compound Annual Growth Rate (CAGR) for the forecast period 2025-2033 is estimated at xx percent, with the market size projected to reach over xx million by 2033, up from xx million in the base year of 2025. Key growth drivers include the increasing demand for advanced prosthetics, advanced drug delivery systems, and sophisticated cardiovascular implants, all of which heavily rely on high-performance polymers. Technological disruptions are primarily centered around the development of smart polymers that can respond to physiological stimuli, actively participate in tissue regeneration, or release therapeutic agents in a controlled manner. Biodegradable and bioresorbable polymers are gaining significant traction as they eliminate the need for a second surgery for implant removal, enhancing patient comfort and reducing healthcare costs. Consumer preferences are shifting towards materials that offer superior biocompatibility, reduced inflammatory responses, and longer functional lifetimes. The competitive dynamics are intense, with established chemical giants competing alongside specialized biomaterial manufacturers. Companies are focusing on vertical integration to control the supply chain and ensure the quality and consistency of their polymeric materials. Market penetration is steadily increasing across developed economies, while emerging markets present substantial untapped potential due to improving healthcare infrastructure and rising disposable incomes. The strategic emphasis on sustainable materials and eco-friendly manufacturing processes is also becoming a significant differentiator in the market.
Dominant Markets & Segments in Polymers For Implantable Medical Devices
The global polymers for implantable medical devices market is characterized by significant regional dominance, with North America leading in terms of market share and innovation, driven by advanced healthcare infrastructure, high R&D investments, and a strong presence of leading medical device manufacturers. Within North America, the United States commands a substantial portion of the market. The market's dominance is further segmented by product type and application, with polymeric materials constituting the largest segment by type, accounting for over xx% of the market share.
Key Drivers of Dominance:
- North America:
- Economic Policies: Favorable government policies supporting healthcare innovation and medical device manufacturing.
- Infrastructure: Well-established healthcare networks, advanced surgical facilities, and a high patient-to-physician ratio.
- R&D Investment: Significant private and public investment in biomedical research and development, fostering innovation in biomaterials.
- Technological Adoption: Early and widespread adoption of advanced medical technologies and implantable devices.
Segment Dominance Analysis:
Application Segments:
- Acetal (POM): This segment holds a significant share due to its excellent mechanical properties, wear resistance, and biocompatibility, making it ideal for applications like joint replacements and dental implants.
- Acrylic (MMA, PMMA): Widely used in intraocular lenses and bone cements, these polymers benefit from their optical clarity and ease of processing. The market for these applications is substantial, driven by the growing demand for ophthalmological and orthopedic procedures.
- Acrylic (hydrogels): Primarily used in contact lenses and soft tissue engineering, hydrogels are experiencing growth due to their high water content and biocompatibility, catering to niche but expanding applications.
- Fluorocarbon: Known for their inertness and chemical resistance, fluoropolymers like PTFE are crucial for vascular grafts and coatings, representing a stable and high-value segment.
- Other: This broad category encompasses a range of specialized polymers, including polyurethanes and silicones, used in diverse applications like catheters, pacemakers, and nerve conduits.
Type Segments:
- Polymeric: As mentioned, this is the dominant type, comprising various polymer families tailored for specific implantable device needs. Its dominance is a testament to the versatility and continuous innovation within polymer science for medical applications.
- Composites: Increasingly important for load-bearing implants, composite materials, often incorporating polymers with reinforcing fibers, offer enhanced strength and stiffness. This segment is poised for significant growth.
- Metallic: While historically dominant, metallic implants are now often complemented or replaced by advanced polymers and composites for certain applications, though they remain critical for high-stress orthopedic implants.
- Ceramic: Used in applications requiring extreme hardness and wear resistance, such as dental implants and some joint components, ceramics represent a niche but critical segment.
- Natural: Polymers derived from natural sources, like collagen and hyaluronic acid, are gaining traction for tissue engineering and regenerative medicine applications, representing an emerging growth area.
The dominance of North America, coupled with the widespread adoption of polymeric materials across various applications, sets the stage for continued market expansion and technological advancement.
Polymers For Implantable Medical Devices Product Developments
Product innovations in implantable medical device polymers are focused on enhancing biocompatibility, achieving controlled degradation profiles, and enabling new functionalities. Key developments include novel polyurethane formulations for flexible cardiovascular implants, advanced polyetheretherketone (PEEK) composites for spinal fusion devices offering superior strength-to-weight ratios, and bioresorbable polyesters designed for controlled drug release in orthopedic applications. Manufacturers are also developing surface modification techniques to improve cell adhesion and reduce the risk of infection. The competitive advantage lies in superior material performance, meeting stringent regulatory standards, and offering tailored solutions for specific medical needs, such as improved lubricity for catheters or enhanced osteoconductivity for bone implants.
Report Scope & Segmentation Analysis
This report meticulously analyzes the global polymers for implantable medical devices market, covering detailed segmentations by application and type. The application segments include Acetal (POM), Acrylic (hydrogels), Acrylic (MMA, PMMA), Fluorocarbon, and Other, each offering unique properties for diverse medical needs. The Type segment encompasses Metallic, Composites, Ceramic, Polymeric, and Natural materials. Each segment is analyzed for its current market size, projected growth rates, key drivers, and competitive dynamics, providing a comprehensive understanding of the market landscape from 2019 to 2033, with a base year of 2025.
- Acetal (POM): Expected to exhibit a CAGR of xx% from 2025-2033, driven by orthopedic and dental applications.
- Acrylic (hydrogels): Projected for a CAGR of xx%, fueled by advancements in ophthalmology and tissue engineering.
- Acrylic (MMA, PMMA): Anticipated to grow at xx% CAGR, supported by demand for intraocular lenses and bone cements.
- Fluorocarbon: Forecasted to grow at xx% CAGR, due to its use in long-term implants like vascular grafts.
- Other: This diverse segment is projected to grow at xx% CAGR, driven by innovations in polyurethanes and silicones.
- Metallic: While a mature segment, it is expected to maintain a CAGR of xx%, essential for high-stress applications.
- Composites: Poised for significant growth with a xx% CAGR, driven by demand for high-performance implants.
- Ceramic: Expected to grow at xx% CAGR, serving specialized applications like dental and some joint replacements.
- Polymeric: This dominant segment is projected to grow at a strong xx% CAGR, underpinning overall market expansion.
- Natural: An emerging segment with a projected CAGR of xx%, driven by regenerative medicine.
Key Drivers of Polymers For Implantable Medical Devices Growth
The growth of the polymers for implantable medical devices market is significantly propelled by several interconnected factors. An aging global population is a primary driver, leading to increased incidence of age-related diseases and a higher demand for joint replacements, cardiovascular devices, and other implants. Technological advancements in medical procedures, particularly the shift towards minimally invasive surgeries, necessitate the development of smaller, more flexible, and biocompatible polymeric components. Furthermore, ongoing research into novel biomaterials, including biodegradable and bioresorbable polymers, is expanding the range of potential applications and improving patient outcomes by reducing the need for revision surgeries. Government initiatives and increasing healthcare spending in emerging economies also play a crucial role, improving access to advanced medical treatments and devices.
Challenges in the Polymers For Implantable Medical Devices Sector
Despite robust growth prospects, the polymers for implantable medical devices sector faces notable challenges. Stringent regulatory approval processes, including extensive preclinical and clinical trials, are time-consuming and costly, posing a significant barrier to market entry for new materials and devices. Supply chain complexities and the need for highly specialized manufacturing capabilities can lead to production bottlenecks and increased costs. The inherent risk of biocompatibility issues and potential long-term adverse reactions necessitates continuous vigilance and advanced material science. Moreover, the competitive pressure from established material suppliers and the evolving landscape of alternative implantable materials demand constant innovation and cost-effectiveness.
Emerging Opportunities in Polymers For Implantable Medical Devices
The polymers for implantable medical devices market presents several promising emerging opportunities. The burgeoning field of regenerative medicine and tissue engineering is creating demand for novel bio-scaffolds and drug delivery vehicles derived from advanced polymers. The increasing focus on personalized medicine is driving the development of custom-fit and patient-specific implants, often utilizing advanced 3D printing technologies with specialized biocompatible polymers. Furthermore, the development of "smart" polymers capable of sensing physiological changes or releasing therapeutic agents in response to specific stimuli offers significant potential for advanced implantable diagnostics and therapeutics. The growing demand for minimally invasive devices also opens doors for highly flexible and conformable polymeric materials.
Leading Players in the Polymers For Implantable Medical Devices Market
BASF Covestro DuPont Celanese Solvay ExxonMobil DSM Eastman Tekni-Plex Evonik Huntsman Formosa Plastics INEOS HEXPOL Kraton Tianjin Plastics Shanghai New Shanghua
Key Developments in Polymers For Implantable Medical Devices Industry
- 2023: DuPont launches a new line of high-performance bioresorbable polymers for orthopedic fixation devices.
- 2023: Covestro develops novel polyurethane formulations for enhanced flexibility in cardiovascular implants.
- 2023: BASF announces strategic partnerships to accelerate R&D in advanced biomaterials for implantable applications.
- 2022: Solvay introduces next-generation PEEK composites with improved mechanical properties for spinal implants.
- 2022: Tekni-Plex expands its capabilities in sterile packaging solutions for polymeric medical devices.
- 2021: Evonik showcases advancements in biodegradable polymers for controlled drug delivery systems.
- 2021: Eastman develops innovative acrylate-based materials for advanced intraocular lenses.
Strategic Outlook for Polymers For Implantable Medical Devices Market
The strategic outlook for the polymers for implantable medical devices market is overwhelmingly positive, driven by continuous innovation in material science, an aging global demographic, and the ongoing shift towards less invasive medical procedures. Companies that focus on developing biocompatible, biodegradable, and functional polymers tailored for specific unmet medical needs will be well-positioned for growth. Strategic collaborations, mergers, and acquisitions will remain key to consolidating market share and acquiring cutting-edge technologies. Investment in research and development, particularly in areas like regenerative medicine and personalized implants, will be crucial for long-term success. Furthermore, navigating the evolving regulatory landscape and ensuring robust supply chains will be critical for capitalizing on the substantial future market potential.
Polymers For Implantable Medical Devices Segmentation
-
1. Application
- 1.1. Acetal (POM)
- 1.2. Acrylic (hydrogels)
- 1.3. Acrylic (MMA, PMMA)
- 1.4. Fluorocarbon
- 1.5. Other
-
2. Type
- 2.1. Metallic
- 2.2. Composites
- 2.3. Ceramic
- 2.4. Polymeric
- 2.5. Natural
Polymers For Implantable Medical Devices Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
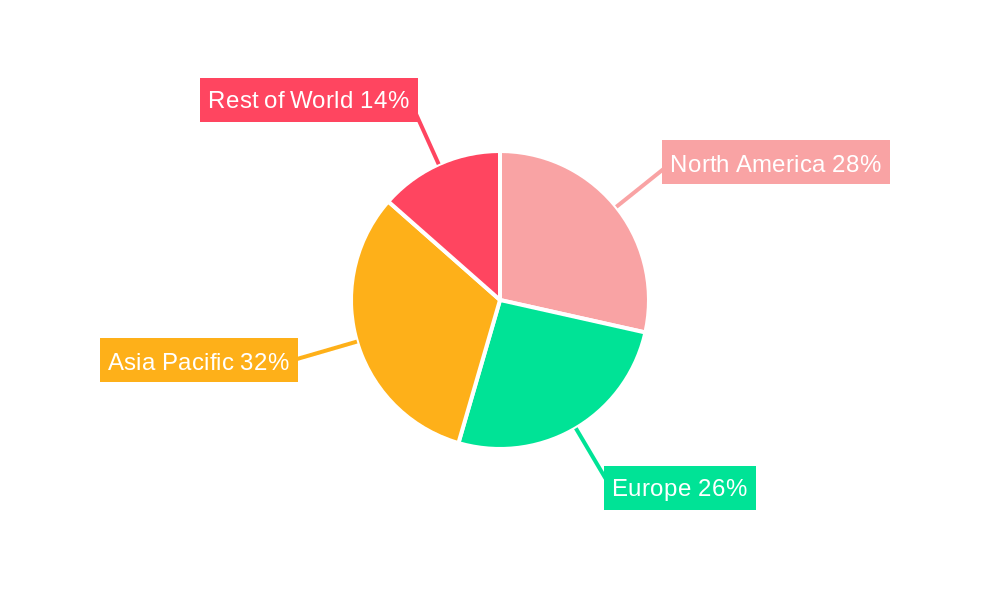
Polymers For Implantable Medical Devices Regional Market Share

Geographic Coverage of Polymers For Implantable Medical Devices
Polymers For Implantable Medical Devices REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 3.8% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. RAX Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Acetal (POM)
- 5.1.2. Acrylic (hydrogels)
- 5.1.3. Acrylic (MMA, PMMA)
- 5.1.4. Fluorocarbon
- 5.1.5. Other
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Metallic
- 5.2.2. Composites
- 5.2.3. Ceramic
- 5.2.4. Polymeric
- 5.2.5. Natural
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Polymers For Implantable Medical Devices Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Acetal (POM)
- 6.1.2. Acrylic (hydrogels)
- 6.1.3. Acrylic (MMA, PMMA)
- 6.1.4. Fluorocarbon
- 6.1.5. Other
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Metallic
- 6.2.2. Composites
- 6.2.3. Ceramic
- 6.2.4. Polymeric
- 6.2.5. Natural
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Polymers For Implantable Medical Devices Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Acetal (POM)
- 7.1.2. Acrylic (hydrogels)
- 7.1.3. Acrylic (MMA, PMMA)
- 7.1.4. Fluorocarbon
- 7.1.5. Other
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Metallic
- 7.2.2. Composites
- 7.2.3. Ceramic
- 7.2.4. Polymeric
- 7.2.5. Natural
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Polymers For Implantable Medical Devices Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Acetal (POM)
- 8.1.2. Acrylic (hydrogels)
- 8.1.3. Acrylic (MMA, PMMA)
- 8.1.4. Fluorocarbon
- 8.1.5. Other
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Metallic
- 8.2.2. Composites
- 8.2.3. Ceramic
- 8.2.4. Polymeric
- 8.2.5. Natural
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Polymers For Implantable Medical Devices Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Acetal (POM)
- 9.1.2. Acrylic (hydrogels)
- 9.1.3. Acrylic (MMA, PMMA)
- 9.1.4. Fluorocarbon
- 9.1.5. Other
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Metallic
- 9.2.2. Composites
- 9.2.3. Ceramic
- 9.2.4. Polymeric
- 9.2.5. Natural
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Polymers For Implantable Medical Devices Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Acetal (POM)
- 10.1.2. Acrylic (hydrogels)
- 10.1.3. Acrylic (MMA, PMMA)
- 10.1.4. Fluorocarbon
- 10.1.5. Other
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Metallic
- 10.2.2. Composites
- 10.2.3. Ceramic
- 10.2.4. Polymeric
- 10.2.5. Natural
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Polymers For Implantable Medical Devices Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Acetal (POM)
- 11.1.2. Acrylic (hydrogels)
- 11.1.3. Acrylic (MMA, PMMA)
- 11.1.4. Fluorocarbon
- 11.1.5. Other
- 11.2. Market Analysis, Insights and Forecast - by Type
- 11.2.1. Metallic
- 11.2.2. Composites
- 11.2.3. Ceramic
- 11.2.4. Polymeric
- 11.2.5. Natural
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 BASF
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Covestro
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 DuPont
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Celanese
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Solvay
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 ExxonMobil
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 DSM
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Eastman
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Tekni-Plex
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Evonik
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Huntsman
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Formosa Plastics
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 INEOS
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.14 HEXPOL
- 12.1.14.1. Company Overview
- 12.1.14.2. Products
- 12.1.14.3. Company Financials
- 12.1.14.4. SWOT Analysis
- 12.1.15 Kraton
- 12.1.15.1. Company Overview
- 12.1.15.2. Products
- 12.1.15.3. Company Financials
- 12.1.15.4. SWOT Analysis
- 12.1.16 Tianjin Plastics
- 12.1.16.1. Company Overview
- 12.1.16.2. Products
- 12.1.16.3. Company Financials
- 12.1.16.4. SWOT Analysis
- 12.1.17 Shanghai New Shanghua
- 12.1.17.1. Company Overview
- 12.1.17.2. Products
- 12.1.17.3. Company Financials
- 12.1.17.4. SWOT Analysis
- 12.1.1 BASF
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Polymers For Implantable Medical Devices Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America Polymers For Implantable Medical Devices Revenue (million), by Application 2025 & 2033
- Figure 3: North America Polymers For Implantable Medical Devices Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Polymers For Implantable Medical Devices Revenue (million), by Type 2025 & 2033
- Figure 5: North America Polymers For Implantable Medical Devices Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Polymers For Implantable Medical Devices Revenue (million), by Country 2025 & 2033
- Figure 7: North America Polymers For Implantable Medical Devices Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Polymers For Implantable Medical Devices Revenue (million), by Application 2025 & 2033
- Figure 9: South America Polymers For Implantable Medical Devices Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Polymers For Implantable Medical Devices Revenue (million), by Type 2025 & 2033
- Figure 11: South America Polymers For Implantable Medical Devices Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Polymers For Implantable Medical Devices Revenue (million), by Country 2025 & 2033
- Figure 13: South America Polymers For Implantable Medical Devices Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Polymers For Implantable Medical Devices Revenue (million), by Application 2025 & 2033
- Figure 15: Europe Polymers For Implantable Medical Devices Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Polymers For Implantable Medical Devices Revenue (million), by Type 2025 & 2033
- Figure 17: Europe Polymers For Implantable Medical Devices Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Polymers For Implantable Medical Devices Revenue (million), by Country 2025 & 2033
- Figure 19: Europe Polymers For Implantable Medical Devices Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Polymers For Implantable Medical Devices Revenue (million), by Application 2025 & 2033
- Figure 21: Middle East & Africa Polymers For Implantable Medical Devices Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Polymers For Implantable Medical Devices Revenue (million), by Type 2025 & 2033
- Figure 23: Middle East & Africa Polymers For Implantable Medical Devices Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Polymers For Implantable Medical Devices Revenue (million), by Country 2025 & 2033
- Figure 25: Middle East & Africa Polymers For Implantable Medical Devices Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Polymers For Implantable Medical Devices Revenue (million), by Application 2025 & 2033
- Figure 27: Asia Pacific Polymers For Implantable Medical Devices Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Polymers For Implantable Medical Devices Revenue (million), by Type 2025 & 2033
- Figure 29: Asia Pacific Polymers For Implantable Medical Devices Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Polymers For Implantable Medical Devices Revenue (million), by Country 2025 & 2033
- Figure 31: Asia Pacific Polymers For Implantable Medical Devices Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Type 2020 & 2033
- Table 3: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Region 2020 & 2033
- Table 4: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Application 2020 & 2033
- Table 5: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Type 2020 & 2033
- Table 6: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Country 2020 & 2033
- Table 7: United States Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 8: Canada Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 9: Mexico Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Application 2020 & 2033
- Table 11: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Type 2020 & 2033
- Table 12: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Country 2020 & 2033
- Table 13: Brazil Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: Argentina Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Application 2020 & 2033
- Table 17: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Type 2020 & 2033
- Table 18: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Germany Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 21: France Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Italy Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 23: Spain Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 24: Russia Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 25: Benelux Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: Nordics Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Application 2020 & 2033
- Table 29: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Type 2020 & 2033
- Table 30: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Country 2020 & 2033
- Table 31: Turkey Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Israel Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 33: GCC Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: North Africa Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 35: South Africa Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 37: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Application 2020 & 2033
- Table 38: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Type 2020 & 2033
- Table 39: Global Polymers For Implantable Medical Devices Revenue million Forecast, by Country 2020 & 2033
- Table 40: China Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 41: India Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Japan Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 43: South Korea Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 45: Oceania Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Polymers For Implantable Medical Devices Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Polymers For Implantable Medical Devices?
The projected CAGR is approximately 3.8%.
2. Which companies are prominent players in the Polymers For Implantable Medical Devices?
Key companies in the market include BASF, Covestro, DuPont, Celanese, Solvay, ExxonMobil, DSM, Eastman, Tekni-Plex, Evonik, Huntsman, Formosa Plastics, INEOS, HEXPOL, Kraton, Tianjin Plastics, Shanghai New Shanghua.
3. What are the main segments of the Polymers For Implantable Medical Devices?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD 1447 million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4250.00, USD 6375.00, and USD 8500.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Polymers For Implantable Medical Devices," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Polymers For Implantable Medical Devices report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Polymers For Implantable Medical Devices?
To stay informed about further developments, trends, and reports in the Polymers For Implantable Medical Devices, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
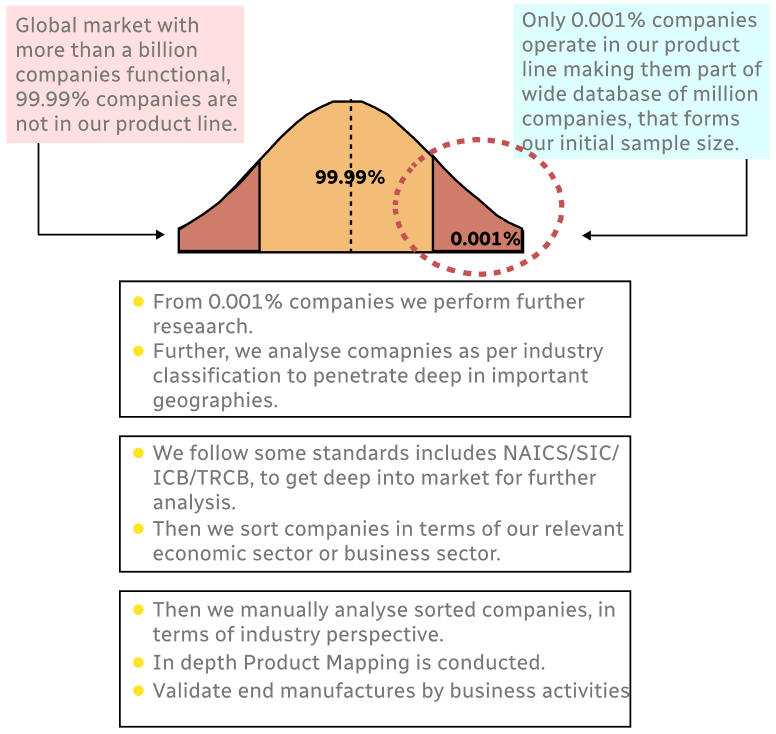
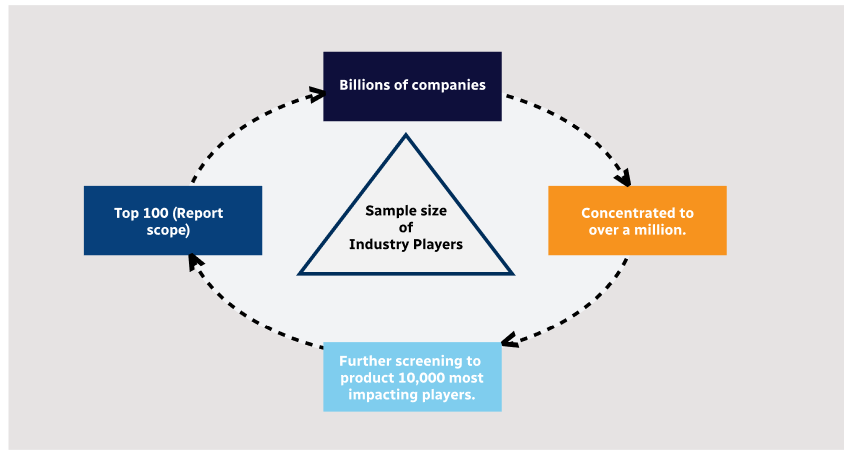
Step 1 - Identification of Relevant Samples Size from Population Database
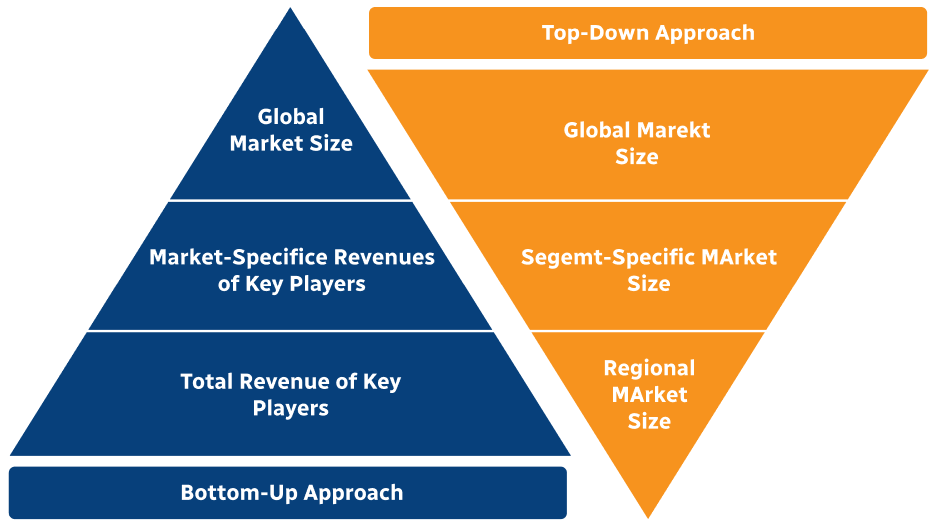
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence