Key Insights
The Neonatal Intensive Care Respiratory Devices market, a crucial segment within the broader neonatal care industry, is experiencing robust growth, driven by several key factors. The increasing prevalence of premature births and low birth weight infants, coupled with advancements in respiratory support technologies, are primary contributors to this expansion. Technological innovations, such as the development of smaller, less invasive, and more efficient devices like high-frequency oscillatory ventilation (HFOV) and non-invasive ventilation (NIV) systems, are improving patient outcomes and driving market demand. Furthermore, the rising incidence of respiratory distress syndrome (RDS) and other neonatal respiratory illnesses fuels the need for advanced respiratory support. The market is also witnessing a shift towards home-based care, fueled by increasing healthcare costs and a preference for convenient, personalized treatment options. This trend necessitates the development of portable and user-friendly devices, creating further opportunities for market expansion. Finally, stringent regulatory frameworks and rising healthcare expenditure in developed and developing nations are also contributing factors. The global market is expected to continue a steady growth trajectory, with a projected compound annual growth rate (CAGR) influenced by ongoing technological innovations and increasing healthcare investments.
The competitive landscape is characterized by a mix of established multinational corporations and specialized smaller companies. Key players are constantly striving for innovation, expanding their product portfolios, and focusing on strategic partnerships and acquisitions to strengthen their market position. Geographic segmentation reveals strong growth in North America and Europe, driven by advanced healthcare infrastructure and high adoption rates of technologically advanced devices. However, emerging economies in Asia-Pacific and other regions are witnessing significant growth potential due to increasing awareness, rising healthcare expenditure, and improving healthcare infrastructure. Regional variations in market dynamics are influenced by factors such as healthcare policies, reimbursement mechanisms, and the level of technological adoption. Challenges remain, such as the high cost of advanced respiratory devices, the need for skilled healthcare professionals, and ensuring equitable access to these life-saving technologies, particularly in resource-constrained settings. Overcoming these challenges will be essential for achieving sustainable growth in the Neonatal Intensive Care Respiratory Devices market in the years to come.
Neonatal Intensive Care Respiratory Devices Industry: A Comprehensive Market Report (2019-2033)
This comprehensive report provides an in-depth analysis of the Neonatal Intensive Care Respiratory Devices industry, offering valuable insights for stakeholders including manufacturers, investors, and healthcare professionals. The report covers market size, growth drivers, challenges, key players, and future trends, utilizing data from the historical period (2019-2024), base year (2025), and forecast period (2025-2033). The total market value is projected to reach xx Million by 2033.
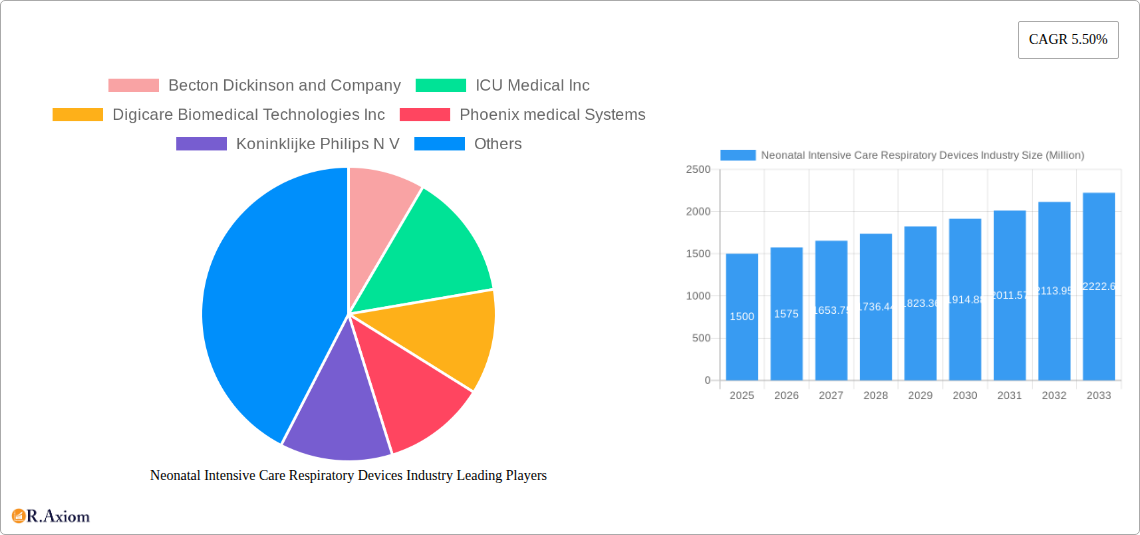
Neonatal Intensive Care Respiratory Devices Industry Market Concentration & Innovation
The Neonatal Intensive Care Respiratory Devices market exhibits a moderately concentrated structure, with several multinational corporations holding significant market share. Leading players like Becton Dickinson and Company, Becton Dickinson and Company, ICU Medical Inc, Koninklijke Philips N V, and Medtronic plc, compete intensely, driving innovation in product design and functionality. Market share data indicates that the top five players collectively account for approximately xx% of the global market in 2025, with Becton Dickinson and Company holding the largest share at approximately xx%. However, smaller companies and emerging players are continuously introducing new technologies and therapies, fostering competition.
Regulatory frameworks, particularly those related to device safety and efficacy, significantly impact market dynamics. The stringent regulatory requirements drive the need for substantial investment in research and development, leading to continuous product innovations. Substitutes for certain devices are limited due to the specialized nature of neonatal care; however, advancements in non-invasive respiratory support and telehealth technologies are emerging as potential alternatives in certain applications. The increasing prevalence of premature births and respiratory illnesses in newborns fuels demand, while mergers and acquisitions (M&A) activities, such as xx Million deals in the past five years, are reshaping the competitive landscape. These acquisitions often involve smaller companies with innovative technologies being integrated into the larger corporations' product portfolios.
Neonatal Intensive Care Respiratory Devices Industry Industry Trends & Insights
The Neonatal Intensive Care Respiratory Devices market is characterized by robust growth, driven by increasing preterm births, rising prevalence of respiratory distress syndrome, and advancements in medical technology. The market is expected to register a CAGR of xx% during the forecast period (2025-2033), reaching a value of xx Million by 2033. Technological advancements such as the integration of smart sensors, improved data analytics, and remote monitoring capabilities are significantly influencing consumer preferences towards technologically advanced, user-friendly devices. This is particularly true in developed economies, where market penetration is already high, and consumers are actively seeking more advanced features. The competitive landscape is highly dynamic, with companies actively engaging in new product development, strategic partnerships, and acquisitions to strengthen their market positions. The rising cost of healthcare and insurance reimbursement policies pose some challenges, however, these are partially offset by the increasing adoption of cost-effective and efficient devices. Additionally, the growing awareness among healthcare professionals and parents about the benefits of advanced neonatal care fuels market growth.
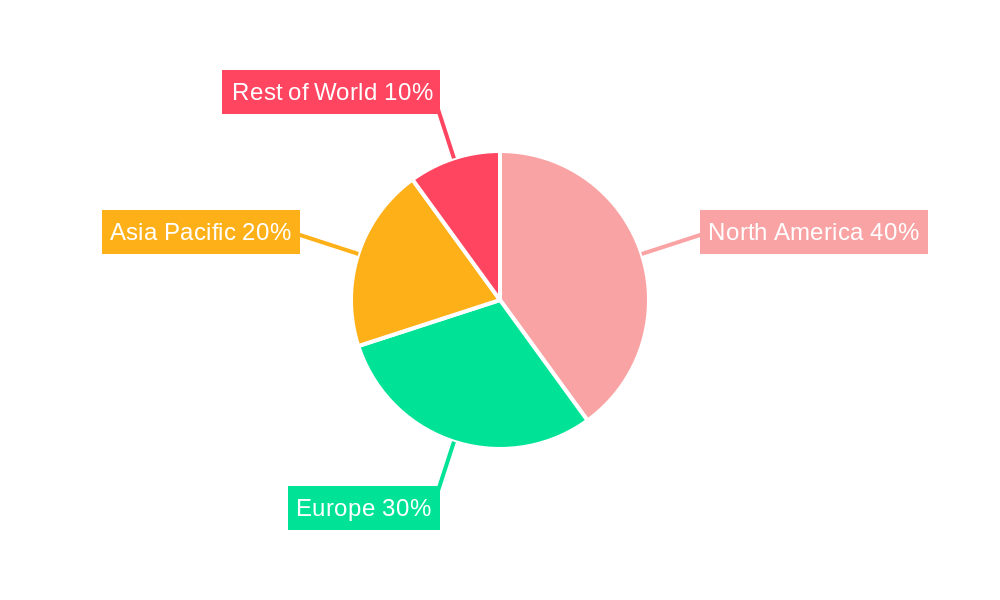
Dominant Markets & Segments in Neonatal Intensive Care Respiratory Devices Industry
North America currently holds the largest market share in the Neonatal Intensive Care Respiratory Devices industry, driven by high healthcare expenditure, advanced healthcare infrastructure, and a significant number of preterm births. Within North America, the United States commands a substantial segment due to factors including:
- High incidence of premature births: A significant number of premature births necessitate advanced respiratory support devices.
- Advanced healthcare infrastructure: Extensive healthcare infrastructure and high levels of technological adoption facilitate the rapid adoption of newer respiratory devices.
- Strong regulatory framework: Well-defined regulatory standards and robust approvals processes ensure the safety and effectiveness of medical devices.
- High healthcare expenditure: The high level of healthcare expenditure allows for greater investment in advanced neonatal care technologies.
The Respiratory Devices segment holds the largest share within the product category, closely followed by Infant Warmers and Incubators, driven by the crucial role these play in maintaining optimal thermal conditions for newborns. Other segments, such as Neonatal Monitoring Devices and Phototherapy Equipment, are also experiencing significant growth, owing to technological advancements and the increasing need for comprehensive neonatal care.
Neonatal Intensive Care Respiratory Devices Industry Product Developments
Recent product innovations focus on miniaturization, portability, and enhanced functionalities. New respiratory devices are integrating advanced sensors and data analytics for real-time monitoring and improved patient outcomes. The trend towards non-invasive ventilation techniques and the development of less-invasive catheters are also noteworthy. These advancements improve patient comfort, reduce the risk of complications, and enhance the overall efficiency of neonatal care. The market is witnessing a push towards more user-friendly interfaces and integrated systems, simplifying the operation and data management for healthcare professionals.
Report Scope & Segmentation Analysis
This report segments the Neonatal Intensive Care Respiratory Devices market by product:
- Infant Warmers: This segment projects xx% CAGR, driven by increasing demand for temperature-controlled environments for newborns. Competition is intense, with companies focusing on energy efficiency and ease of use.
- Incubators: The incubator market is expected to grow at a CAGR of xx%, driven by technological advancements, such as improved humidity and oxygen control. Competition is focused on innovation in materials and design to enhance portability and ease of cleaning.
- Neonatal Monitoring Devices: This segment is projected to witness a CAGR of xx%, driven by the growing need for real-time data monitoring and remote patient monitoring capabilities. Competition centers on advanced features and integration with hospital information systems.
- Respiratory Devices: This segment, the largest by market share, is anticipated to grow at a CAGR of xx%, driven by the rising incidence of respiratory disorders in neonates and advancements in ventilation technologies. Competition focuses on the development of advanced features, such as non-invasive ventilation options and improved patient safety mechanisms.
- Phototherapy Equipment: The phototherapy equipment segment is projected to grow at a CAGR of xx%, driven by the increasing incidence of hyperbilirubinemia in newborns. Competition is centered on the efficiency and safety of the equipment.
- Catheters: This segment's growth is projected at xx% CAGR, driven by advances in material science that allow for smaller and less invasive catheters. Competition is centered around catheter design, material, and sterility.
- Other Products: This category includes ancillary devices and supplies; it's expected to grow at a CAGR of xx%.
Key Drivers of Neonatal Intensive Care Respiratory Devices Growth
The growth of this industry is propelled by several factors: the rising prevalence of premature births and respiratory illnesses in newborns; technological advancements leading to smaller, more efficient, and user-friendly devices; increasing healthcare expenditure globally, particularly in developing economies; and supportive regulatory frameworks encouraging innovation and adoption of advanced technologies. Further, growing awareness among healthcare professionals and parents about the importance of advanced neonatal care plays a vital role.
Challenges in the Neonatal Intensive Care Respiratory Devices Industry Sector
Challenges include the high cost of advanced devices limiting accessibility in low-resource settings; stringent regulatory requirements and lengthy approval processes increasing development costs and timelines; supply chain disruptions impacting device availability and pricing; intense competition from established players and the emergence of new market entrants. The variability in reimbursement policies across different healthcare systems also presents a significant challenge.
Emerging Opportunities in Neonatal Intensive Care Respiratory Devices Industry
Emerging opportunities lie in the development of portable and wireless monitoring devices, the integration of artificial intelligence and machine learning for predictive analytics and personalized treatment, expansion into emerging markets with unmet needs, and strategic partnerships to improve device access and affordability. The development of more sustainable and eco-friendly devices also presents a significant opportunity.
Leading Players in the Neonatal Intensive Care Respiratory Devices Industry Market
- Becton Dickinson and Company
- ICU Medical Inc
- Digicare Biomedical Technologies Inc
- Phoenix medical Systems
- Koninklijke Philips N V
- Cook Group (Cook Medical)
- Air Liquide Medical Systems India
- B Braun Melsungen AG
- Getinge AB
- Teleflex Incorporated
- Medtronic plc
- Utah Medical Products Inc
- Drägerwerk AG & Co KGaA
Key Developments in Neonatal Intensive Care Respiratory Devices Industry
- June 2022: Dräger received Frost & Sullivan's 2022 Global New Product Innovation Leadership Award for neonatal and respiratory care. This highlights innovation in the sector and strengthens Dräger's market position.
- May 2022: Pinnacle Industries launched a new range of neonatal ambulances for the Maharashtra Government in India. This signifies increased investment in neonatal transportation and care infrastructure, boosting market demand.
Strategic Outlook for Neonatal Intensive Care Respiratory Devices Market
The Neonatal Intensive Care Respiratory Devices market presents a strong growth outlook, driven by continued technological advancements, rising healthcare expenditure, and growing awareness of the importance of neonatal care. Further market expansion is expected in emerging economies as healthcare infrastructure improves and access to advanced medical technologies increases. Companies focusing on innovation, strategic partnerships, and cost-effective solutions are poised to gain a competitive edge in this dynamic market.
Neonatal Intensive Care Respiratory Devices Industry Segmentation
-
1. Product
- 1.1. Infant Warmers
- 1.2. Incubators
- 1.3. Neonatal Monitoring Devices
- 1.4. Respiratory Devices
- 1.5. Phototherapy Equipment
- 1.6. Catheters
- 1.7. Other Products
Neonatal Intensive Care Respiratory Devices Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
Neonatal Intensive Care Respiratory Devices Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2019-2033 |
| Base Year | 2024 |
| Estimated Year | 2025 |
| Forecast Period | 2025-2033 |
| Historical Period | 2019-2024 |
| Growth Rate | CAGR of 5.50% from 2019-2033 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.2.1. Rising Incidence of Preterm Births; Increasing Prevalence of Chronic Diseases Among the Newborn Babies; Technological Advancements in Neonatal Intensive Care Devices
- 3.3. Market Restrains
- 3.3.1. Lack of Awareness and Economic Constraints in Developing Countries; Decreasing Birth Rates in Developed Countries
- 3.4. Market Trends
- 3.4.1. Incubators Segment is Expected to Cover a Significant Share Over the Forecast Period
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Neonatal Intensive Care Respiratory Devices Industry Analysis, Insights and Forecast, 2019-2031
- 5.1. Market Analysis, Insights and Forecast - by Product
- 5.1.1. Infant Warmers
- 5.1.2. Incubators
- 5.1.3. Neonatal Monitoring Devices
- 5.1.4. Respiratory Devices
- 5.1.5. Phototherapy Equipment
- 5.1.6. Catheters
- 5.1.7. Other Products
- 5.2. Market Analysis, Insights and Forecast - by Region
- 5.2.1. North America
- 5.2.2. Europe
- 5.2.3. Asia Pacific
- 5.2.4. Middle East and Africa
- 5.2.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Product
- 6. North America Neonatal Intensive Care Respiratory Devices Industry Analysis, Insights and Forecast, 2019-2031
- 6.1. Market Analysis, Insights and Forecast - by Product
- 6.1.1. Infant Warmers
- 6.1.2. Incubators
- 6.1.3. Neonatal Monitoring Devices
- 6.1.4. Respiratory Devices
- 6.1.5. Phototherapy Equipment
- 6.1.6. Catheters
- 6.1.7. Other Products
- 6.1. Market Analysis, Insights and Forecast - by Product
- 7. Europe Neonatal Intensive Care Respiratory Devices Industry Analysis, Insights and Forecast, 2019-2031
- 7.1. Market Analysis, Insights and Forecast - by Product
- 7.1.1. Infant Warmers
- 7.1.2. Incubators
- 7.1.3. Neonatal Monitoring Devices
- 7.1.4. Respiratory Devices
- 7.1.5. Phototherapy Equipment
- 7.1.6. Catheters
- 7.1.7. Other Products
- 7.1. Market Analysis, Insights and Forecast - by Product
- 8. Asia Pacific Neonatal Intensive Care Respiratory Devices Industry Analysis, Insights and Forecast, 2019-2031
- 8.1. Market Analysis, Insights and Forecast - by Product
- 8.1.1. Infant Warmers
- 8.1.2. Incubators
- 8.1.3. Neonatal Monitoring Devices
- 8.1.4. Respiratory Devices
- 8.1.5. Phototherapy Equipment
- 8.1.6. Catheters
- 8.1.7. Other Products
- 8.1. Market Analysis, Insights and Forecast - by Product
- 9. Middle East and Africa Neonatal Intensive Care Respiratory Devices Industry Analysis, Insights and Forecast, 2019-2031
- 9.1. Market Analysis, Insights and Forecast - by Product
- 9.1.1. Infant Warmers
- 9.1.2. Incubators
- 9.1.3. Neonatal Monitoring Devices
- 9.1.4. Respiratory Devices
- 9.1.5. Phototherapy Equipment
- 9.1.6. Catheters
- 9.1.7. Other Products
- 9.1. Market Analysis, Insights and Forecast - by Product
- 10. South America Neonatal Intensive Care Respiratory Devices Industry Analysis, Insights and Forecast, 2019-2031
- 10.1. Market Analysis, Insights and Forecast - by Product
- 10.1.1. Infant Warmers
- 10.1.2. Incubators
- 10.1.3. Neonatal Monitoring Devices
- 10.1.4. Respiratory Devices
- 10.1.5. Phototherapy Equipment
- 10.1.6. Catheters
- 10.1.7. Other Products
- 10.1. Market Analysis, Insights and Forecast - by Product
- 11. North America Neonatal Intensive Care Respiratory Devices Industry Analysis, Insights and Forecast, 2019-2031
- 11.1. Market Analysis, Insights and Forecast - By Country/Sub-region
- 11.1.1 United States
- 11.1.2 Canada
- 11.1.3 Mexico
- 12. Europe Neonatal Intensive Care Respiratory Devices Industry Analysis, Insights and Forecast, 2019-2031
- 12.1. Market Analysis, Insights and Forecast - By Country/Sub-region
- 12.1.1 Germany
- 12.1.2 United Kingdom
- 12.1.3 France
- 12.1.4 Italy
- 12.1.5 Spain
- 12.1.6 Rest of Europe
- 13. Asia Pacific Neonatal Intensive Care Respiratory Devices Industry Analysis, Insights and Forecast, 2019-2031
- 13.1. Market Analysis, Insights and Forecast - By Country/Sub-region
- 13.1.1 China
- 13.1.2 Japan
- 13.1.3 India
- 13.1.4 Australia
- 13.1.5 South Korea
- 13.1.6 Rest of Asia Pacific
- 14. Middle East and Africa Neonatal Intensive Care Respiratory Devices Industry Analysis, Insights and Forecast, 2019-2031
- 14.1. Market Analysis, Insights and Forecast - By Country/Sub-region
- 14.1.1 GCC
- 14.1.2 South Africa
- 14.1.3 Rest of Middle East and Africa
- 15. South America Neonatal Intensive Care Respiratory Devices Industry Analysis, Insights and Forecast, 2019-2031
- 15.1. Market Analysis, Insights and Forecast - By Country/Sub-region
- 15.1.1 Brazil
- 15.1.2 Argentina
- 15.1.3 Rest of South America
- 16. Competitive Analysis
- 16.1. Global Market Share Analysis 2024
- 16.2. Company Profiles
- 16.2.1 Becton Dickinson and Company
- 16.2.1.1. Overview
- 16.2.1.2. Products
- 16.2.1.3. SWOT Analysis
- 16.2.1.4. Recent Developments
- 16.2.1.5. Financials (Based on Availability)
- 16.2.2 ICU Medical Inc
- 16.2.2.1. Overview
- 16.2.2.2. Products
- 16.2.2.3. SWOT Analysis
- 16.2.2.4. Recent Developments
- 16.2.2.5. Financials (Based on Availability)
- 16.2.3 Digicare Biomedical Technologies Inc
- 16.2.3.1. Overview
- 16.2.3.2. Products
- 16.2.3.3. SWOT Analysis
- 16.2.3.4. Recent Developments
- 16.2.3.5. Financials (Based on Availability)
- 16.2.4 Phoenix medical Systems
- 16.2.4.1. Overview
- 16.2.4.2. Products
- 16.2.4.3. SWOT Analysis
- 16.2.4.4. Recent Developments
- 16.2.4.5. Financials (Based on Availability)
- 16.2.5 Koninklijke Philips N V
- 16.2.5.1. Overview
- 16.2.5.2. Products
- 16.2.5.3. SWOT Analysis
- 16.2.5.4. Recent Developments
- 16.2.5.5. Financials (Based on Availability)
- 16.2.6 Cook Group (Cook Medical)
- 16.2.6.1. Overview
- 16.2.6.2. Products
- 16.2.6.3. SWOT Analysis
- 16.2.6.4. Recent Developments
- 16.2.6.5. Financials (Based on Availability)
- 16.2.7 Air Liquide Medical Systems India
- 16.2.7.1. Overview
- 16.2.7.2. Products
- 16.2.7.3. SWOT Analysis
- 16.2.7.4. Recent Developments
- 16.2.7.5. Financials (Based on Availability)
- 16.2.8 B Braun Melsungen AG
- 16.2.8.1. Overview
- 16.2.8.2. Products
- 16.2.8.3. SWOT Analysis
- 16.2.8.4. Recent Developments
- 16.2.8.5. Financials (Based on Availability)
- 16.2.9 Getinge AB*List Not Exhaustive
- 16.2.9.1. Overview
- 16.2.9.2. Products
- 16.2.9.3. SWOT Analysis
- 16.2.9.4. Recent Developments
- 16.2.9.5. Financials (Based on Availability)
- 16.2.10 Teleflex Incorporated
- 16.2.10.1. Overview
- 16.2.10.2. Products
- 16.2.10.3. SWOT Analysis
- 16.2.10.4. Recent Developments
- 16.2.10.5. Financials (Based on Availability)
- 16.2.11 Medtronic plc
- 16.2.11.1. Overview
- 16.2.11.2. Products
- 16.2.11.3. SWOT Analysis
- 16.2.11.4. Recent Developments
- 16.2.11.5. Financials (Based on Availability)
- 16.2.12 Utah Medical Products Inc
- 16.2.12.1. Overview
- 16.2.12.2. Products
- 16.2.12.3. SWOT Analysis
- 16.2.12.4. Recent Developments
- 16.2.12.5. Financials (Based on Availability)
- 16.2.13 Drgerwerk AG & Co KGaA
- 16.2.13.1. Overview
- 16.2.13.2. Products
- 16.2.13.3. SWOT Analysis
- 16.2.13.4. Recent Developments
- 16.2.13.5. Financials (Based on Availability)
- 16.2.1 Becton Dickinson and Company
List of Figures
- Figure 1: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Breakdown (Million, %) by Region 2024 & 2032
- Figure 2: North America Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Country 2024 & 2032
- Figure 3: North America Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Country 2024 & 2032
- Figure 4: Europe Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Country 2024 & 2032
- Figure 5: Europe Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Country 2024 & 2032
- Figure 6: Asia Pacific Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Country 2024 & 2032
- Figure 7: Asia Pacific Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Country 2024 & 2032
- Figure 8: Middle East and Africa Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Country 2024 & 2032
- Figure 9: Middle East and Africa Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Country 2024 & 2032
- Figure 10: South America Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Country 2024 & 2032
- Figure 11: South America Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Country 2024 & 2032
- Figure 12: North America Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Product 2024 & 2032
- Figure 13: North America Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Product 2024 & 2032
- Figure 14: North America Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Country 2024 & 2032
- Figure 15: North America Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Country 2024 & 2032
- Figure 16: Europe Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Product 2024 & 2032
- Figure 17: Europe Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Product 2024 & 2032
- Figure 18: Europe Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Country 2024 & 2032
- Figure 19: Europe Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Country 2024 & 2032
- Figure 20: Asia Pacific Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Product 2024 & 2032
- Figure 21: Asia Pacific Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Product 2024 & 2032
- Figure 22: Asia Pacific Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Country 2024 & 2032
- Figure 23: Asia Pacific Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Country 2024 & 2032
- Figure 24: Middle East and Africa Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Product 2024 & 2032
- Figure 25: Middle East and Africa Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Product 2024 & 2032
- Figure 26: Middle East and Africa Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Country 2024 & 2032
- Figure 27: Middle East and Africa Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Country 2024 & 2032
- Figure 28: South America Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Product 2024 & 2032
- Figure 29: South America Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Product 2024 & 2032
- Figure 30: South America Neonatal Intensive Care Respiratory Devices Industry Revenue (Million), by Country 2024 & 2032
- Figure 31: South America Neonatal Intensive Care Respiratory Devices Industry Revenue Share (%), by Country 2024 & 2032
List of Tables
- Table 1: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Region 2019 & 2032
- Table 2: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Product 2019 & 2032
- Table 3: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Region 2019 & 2032
- Table 4: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Country 2019 & 2032
- Table 5: United States Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 6: Canada Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 7: Mexico Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 8: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Country 2019 & 2032
- Table 9: Germany Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 10: United Kingdom Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 11: France Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 12: Italy Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 13: Spain Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 14: Rest of Europe Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 15: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Country 2019 & 2032
- Table 16: China Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 17: Japan Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 18: India Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 19: Australia Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 20: South Korea Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 21: Rest of Asia Pacific Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 22: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Country 2019 & 2032
- Table 23: GCC Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 24: South Africa Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 25: Rest of Middle East and Africa Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 26: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Country 2019 & 2032
- Table 27: Brazil Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 28: Argentina Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 29: Rest of South America Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 30: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Product 2019 & 2032
- Table 31: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Country 2019 & 2032
- Table 32: United States Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 33: Canada Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 34: Mexico Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 35: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Product 2019 & 2032
- Table 36: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Country 2019 & 2032
- Table 37: Germany Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 38: United Kingdom Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 39: France Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 40: Italy Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 41: Spain Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 42: Rest of Europe Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 43: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Product 2019 & 2032
- Table 44: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Country 2019 & 2032
- Table 45: China Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 46: Japan Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 47: India Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 48: Australia Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 49: South Korea Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 50: Rest of Asia Pacific Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 51: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Product 2019 & 2032
- Table 52: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Country 2019 & 2032
- Table 53: GCC Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 54: South Africa Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 55: Rest of Middle East and Africa Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 56: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Product 2019 & 2032
- Table 57: Global Neonatal Intensive Care Respiratory Devices Industry Revenue Million Forecast, by Country 2019 & 2032
- Table 58: Brazil Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 59: Argentina Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
- Table 60: Rest of South America Neonatal Intensive Care Respiratory Devices Industry Revenue (Million) Forecast, by Application 2019 & 2032
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Neonatal Intensive Care Respiratory Devices Industry?
The projected CAGR is approximately 5.50%.
2. Which companies are prominent players in the Neonatal Intensive Care Respiratory Devices Industry?
Key companies in the market include Becton Dickinson and Company, ICU Medical Inc, Digicare Biomedical Technologies Inc, Phoenix medical Systems, Koninklijke Philips N V, Cook Group (Cook Medical), Air Liquide Medical Systems India, B Braun Melsungen AG, Getinge AB*List Not Exhaustive, Teleflex Incorporated, Medtronic plc, Utah Medical Products Inc, Drgerwerk AG & Co KGaA.
3. What are the main segments of the Neonatal Intensive Care Respiratory Devices Industry?
The market segments include Product.
4. Can you provide details about the market size?
The market size is estimated to be USD XX Million as of 2022.
5. What are some drivers contributing to market growth?
Rising Incidence of Preterm Births; Increasing Prevalence of Chronic Diseases Among the Newborn Babies; Technological Advancements in Neonatal Intensive Care Devices.
6. What are the notable trends driving market growth?
Incubators Segment is Expected to Cover a Significant Share Over the Forecast Period.
7. Are there any restraints impacting market growth?
Lack of Awareness and Economic Constraints in Developing Countries; Decreasing Birth Rates in Developed Countries.
8. Can you provide examples of recent developments in the market?
In June 2022, Frost & Sullivan selected Dräger for its 2022 Global New Product Innovation Leadership Award in the categories of neonatal and respiratory care.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Neonatal Intensive Care Respiratory Devices Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Neonatal Intensive Care Respiratory Devices Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Neonatal Intensive Care Respiratory Devices Industry?
To stay informed about further developments, trends, and reports in the Neonatal Intensive Care Respiratory Devices Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database


Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence



